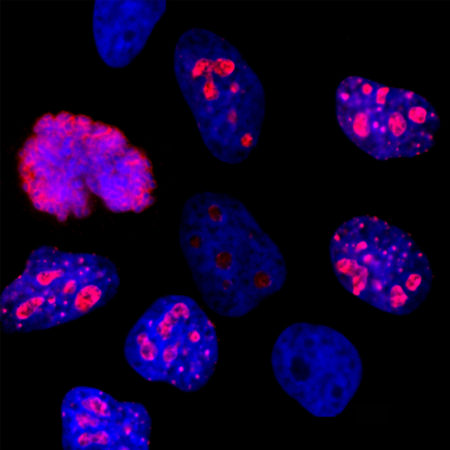
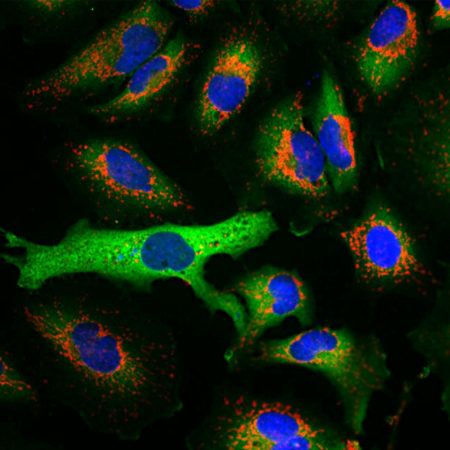
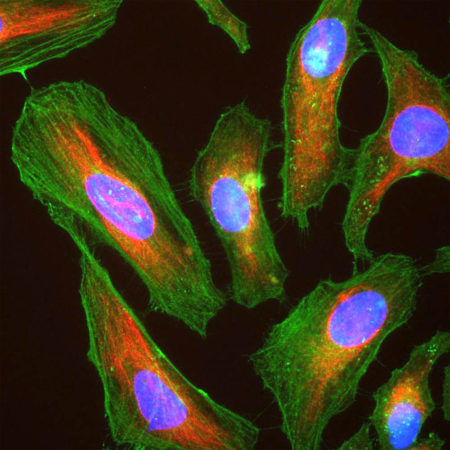
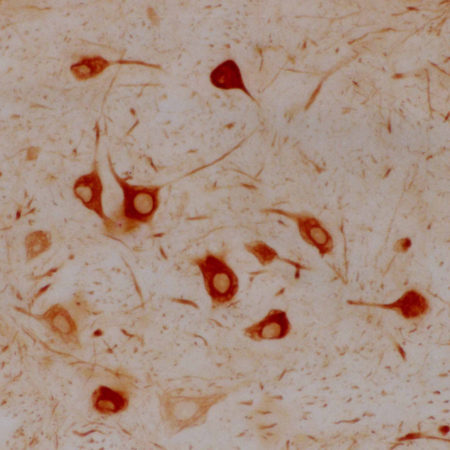
| Name: | Mouse Monoclonal antibody against netrin domain of C3 |
| Immunogen: | Recombinant netrin domain of human C3 construct |
| HGNC Name: | C3 |
| UniProt: | P01024 |
| Molecular Weight: | 185kDa |
| Host: | Mouse |
| Isotype: | IgG1 |
| Species Cross-Reactivity: | Human |
| RRID: | AB_2572257 |
| Format: | Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, ELISA |
| Recommended Dilutions: | WB: 1:1,000-1:5,000. |
| Storage: | Store at 4°C for short term, for longer term at -20°C |
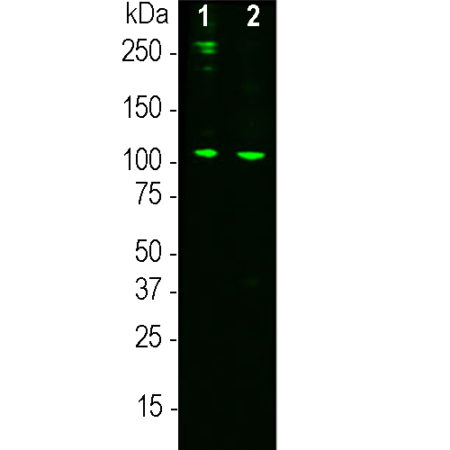
Western blot analysis of MCA-2B5 on human serum samples. 0.1μg human C3 protein (lane 1), 10μg normal human serum proteins (lane 2) was blotted with MCA-2B5 at a concentration 1:3,000. The MCA-2B5 monoclonal binds strongly and cleanly to a band at about 115 kDa which represents the intact α subunit of C3 and a weaker proteolytic band at approximately 45kDa which is the C-terminal of the α subunit including the netrin domain. Bands at 190kDa and above are likely the pro-C3 and its glycosylated form.
Mouse Monoclonal Antibody to Complement C3 α-chain
Cat# MCA-2B5
$120.00 – $800.00
Complement component 3, often simply called C3, is the third protein in the complement system. The complement system “complements” the ability of antibodies and phagocytic cells to clear pathogens such as bacteria and viruses from the organism and can trigger inflammation and remove debris from cells and tissues. C3 is involved in both the classical and alternative pathway. The MCA-2B5 antibody binds to the netrin-lke domain in the α subunit of human C3.
Complement component 3, often simply called C3, is the third protein in the complement system. The complement system “complements” the ability of antibodies and phagocytic cells to clear pathogens such as bacteria and viruses from the organism, can trigger inflammation and remove debris from cells and tissues (1). C3 is involved in both the classical and alternative pathway. C3 is synthesized as an intracellular pro-C3 precursor molecule of 185 kDa which is processed by proteolytic cleavage into two large chains the C-terminal α subunit (115 kDa) and the N-terminal β subunit (70 kDa). The two chains however remain linked by a disulfide bond (2).
C3 activation involves further cleavage by the enzyme C3 convertase to produce C3a (9 kDa derived from the N-terminus of the α subunit) and C3b (the rest of the molecule including the β subunit and truncated α subunit). C3a is released into the surrounding fluids and can bind to receptors on basophils and mast cells triggering them to release histamine and other vasoactive components. Because of the role of these materials in anaphylaxis, C3a is called an anaphylatoxin, and the N-terminal region of the α subunit is referred to as the anaphyloxin domain. EnCor markets a human recombinant form of this domain and has made antibodies to it. C3a may be further cleaved by carboxypeptidase to produce C3adesArg or ASP (acylation-stimulating protein), which acts as a paracrine signal to increase triglyceride synthesis in adipocytes (3). C3adesArg has been demonstrated to be present at increased levels in patients with obesity, type 2 diabetes and coronary artery disease (4,5,6). C3b is the main effector molecule of the complement system, expressing multiple binding sites for other complement components such as C5, properdin, factor B, factor H and certain membrane proteins such as MCP. Binding these proteins to C3b leads either to amplification of C3 convertase, or initiation of the Membrane Attack Complex (MAC) of proteins. C3b also serves as an opsonizing agent to bind to the pathogen and target it for destruction by phagocytes. Overall, C3 promotes phagocytosis, supports local inflammatory responses against pathogens, and instructs the adaptive immune response to select the appropriate antigens for a humoral response (6). More recently, C3 has been suggested to have a pathophysiological role in Alzheimer’s and other neurodegenerative disorders (7). In clinical practice the level of C3 in serum and CSF can be used to help identify immunological disorders, especially those associated with deficiencies of complement components.
1. Janeway CA et al. The complement system and innate immunity. The Immune System in Health and Disease. 5th edition (2001).
2. Brade V, Hall RE, Colten HR. Biosynthesis of pro-C3, a precursor of the third component of complement.J. Exp. Med. 146:759-765 (1977).
3. Xia Z, ET AL. Acylation-stimulating Protein (ASP)/Complement C3adesArg Deficiency Results in Increased Energy Expenditure in Mice. J. Biol. Chem. 279:4051-7 (2004).
4. Maslowska M, et al. Plasma acylation stimulating protein, adipsin and lipids in non-obese and obese populations. Eur. J. Clin. Inv. 29:679–86 (1999).
5. Koistinen HA, et al. Plasma Acylation Stimulating Protein Concentration and Subcutaneous Adipose Tissue C3 mRNA Expression in Nondiabetic and Type 2 Diabetic Men. Arteriosclerosis, Thrombosis, and Vascular Biology 21:1034–9 (2001).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com