| Name: | Chicken Polyclonal Antibody to Calbindin |
| Immunogen: | Full-length recombinant human protein expressed in and purified from E. coli. |
| HGNC Name: | CALB1 |
| UniProt: | P05937 |
| Molecular Weight: | 28kDa |
| Host: | Chicken |
| Isotype: | |
| Species Cross-Reactivity: | Human, cow, rat, mouse |
| RRID: | AB_2572237 |
| Format: | Concentrated IgY preparation plus 0.02% NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:5,000. IF/ICC 1:1,000-1:5,000 or IHC: 1;10,000 |
| Storage: | Stable at 4°C for 1 year. |
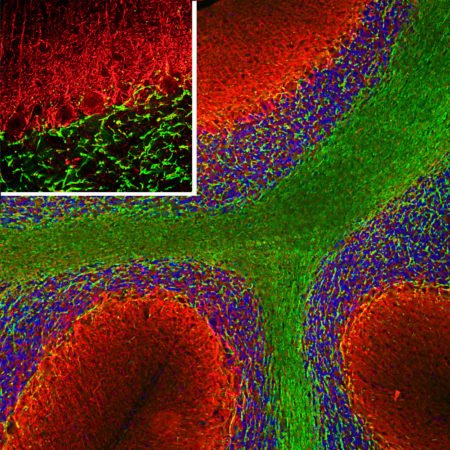
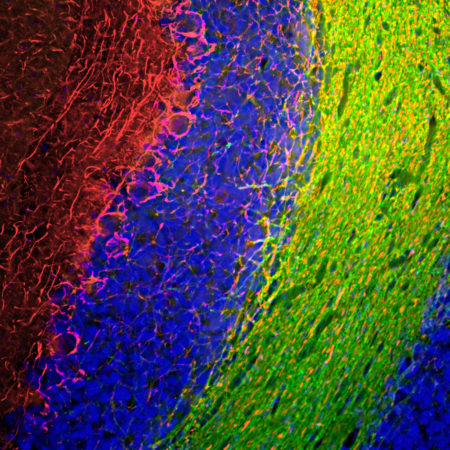
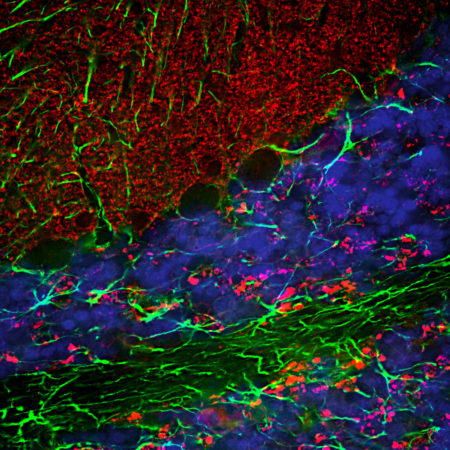
Immunofluorescent analysis of rat cerebellum section stained with chicken pAb to calbindin, CPCA-Calb, dilution 1:2,000, in green, and costained with rabbit pAb to MeCP2, RPCA-MeCP2, dilution 1:5,000, in red. Following transcardial perfusion with 4% paraformaldehyde, brain was post fixed for 24 hours, cut to 45μM, and free-floating sections were stained with above antibodies. Calbindin, often used as a Purkinje cell marker, is prominently detected in dendrites and perikarya of these cells in the cerebellar molecular layer. The MeCP2 antibody selectively stains nuclei of neuronal cells.
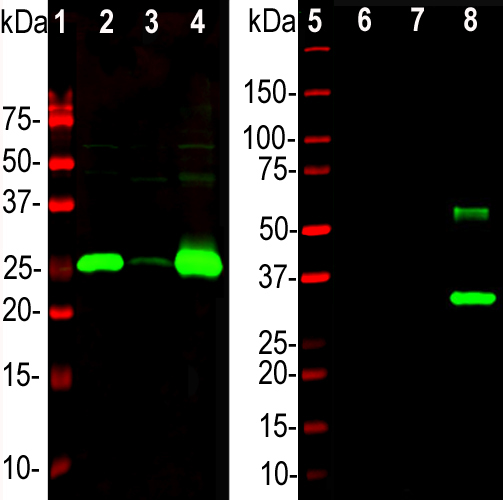
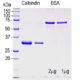
Western blot analysis of different tissue lysates and recombinant protein solutions using chicken pAb to calbindin, CPCA-Calb, dilution 1:5,000 in green: [1] protein standard (red), [2] rat cerebellum, [3] pig hippocampus, [4] cow cerebellum, [5] protein standard (red). Next lanes are full length recombinant human proteins, [6] parvalbumin, [7] calretinin, [8] calbindin. Bands at ~25kDa in tissue lysates and ~30 kDa in protein solutions correspond to native calbindin, the recombinant forms of calbindin, caretinin and parvalbumin being slightly larger due to the presence of a His tag and other vector derived sequence. The CPCA-Calb antibody specifically recognizes calbindin protein and does not react with the closely related proteins parvalbumin and calretinin.
Chicken Polyclonal Antibody to Calbindin
Cat# CPCA-Calb
$120.00 – $800.00
Calbindin, also known as calbindin 1 or calbindin-D28k is a member of the large superfamily of cytoplasmic EF hand containing Calcium binding proteins and is expressed in the brain, intestine, kidney and pancreas (1-3). It is particularly concentrated in the dendrites and perikarya of cerebellar Purkinje cells, but is also found in many GABAergic interneurons in the cerebral cortex. These GABAergic interneurons in most cases express only one of three Calcium binding proteins, namely calbindin or parvalbumin or calretinin. As a result these important and physiologically distinct inhibitory interneurons can be identified and subclassified based on their content of these three proteins (4-6).
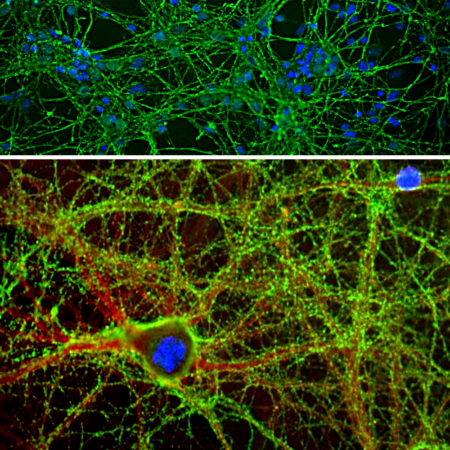
The CPCA-Calb antibody was made against full length recombinant human calbindin expressed in and purified from E. coli, EnCor product Prot-r-Calb1. We have shown that the antibody binds calbindin cleanly on western blots and in sections but fails to recognize the related calretinin and parvalbumin proteins. It is therefore ideally suited for identifying and subclassifying cortical GABAergic neurons. EnCor also manufactures mouse monoclonal antibodies to calbindin MCA-4H7 and MCA-5A9. We also supply a variety of other mouse, rabbit and chicken antibodies to parvalbumin (MCA-3C9 and CPCA-Pvalb) and calretinin (MCA-3G9, MCA-6A9 and CPCA-Calret), allowing double and triple labeling of appropriate cells and tissue specimens. Mouse select image above left for larger view.
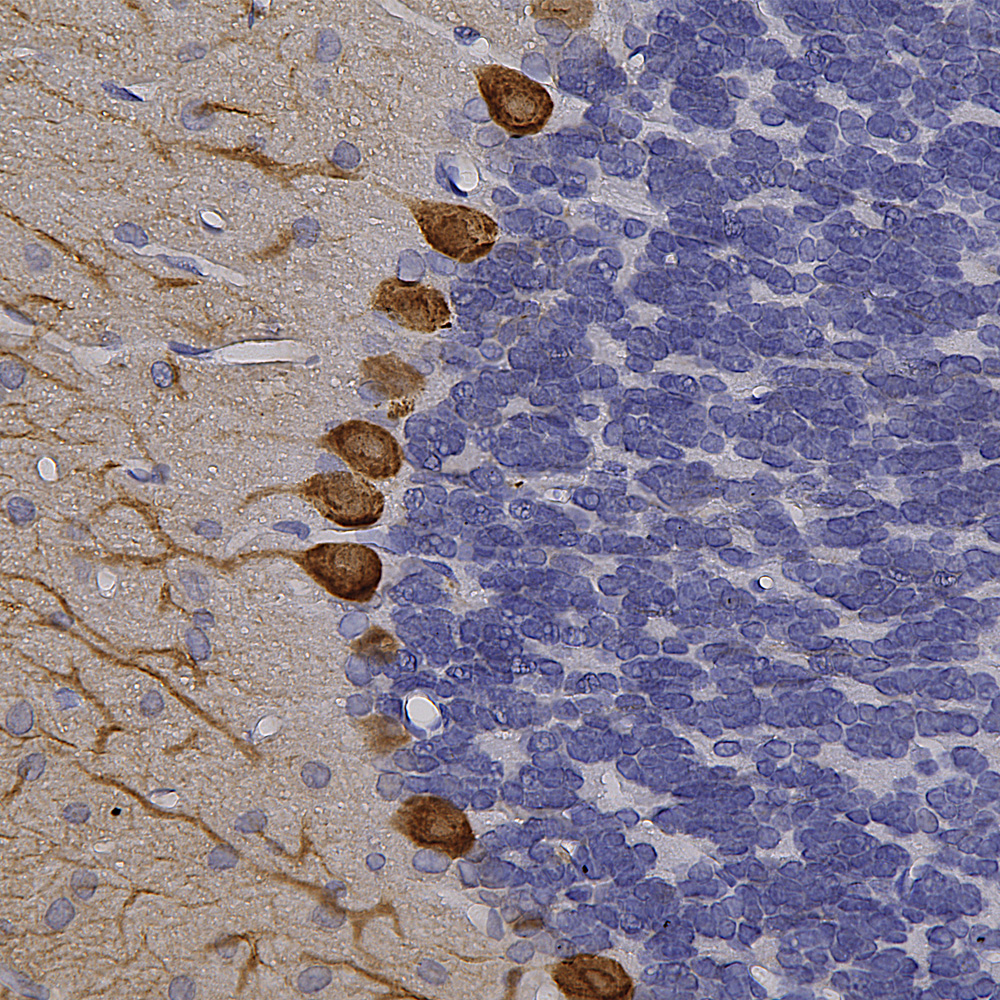
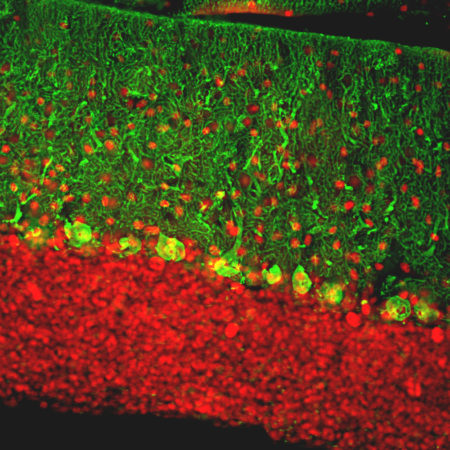
Chromogenic Immunostaining of a formalin fixed paraffin embedded rat cerebellum section with chicken pAb to calbindin, CPCA-Calb, dilution 1:10,000, detected in DAB (brown) following the ABC method. Hematoxylin (blue) was used as the counterstain. Purkinje cells and their dendrites stain prominently with this antibody. Mouse select image for larger view.
1. Kretsinger RH, Nockolds CE. Carp Muscle Calcium-binding Protein: II. Structure determination and general description. J. Biol. Chem. 248:3313-26 (1973).
2. Andressen C, Bliimcke I, Celio MR. Calcium-binding proteins: selective markers of nerve cells. Cell Tissue Res. 271:181-208 (1993).
3. Schwaller B, Meyer M, Schiffmann S. ‘New’ functions for ‘old’ proteins: The role of the calcium binding proteins calbindin D-28k, calretinin and parvalbumin, in cerebellar physiology. Studies with knockout mice. The Cerebellum 1:241–58 (2002).
4. Celio MR. Calbindin D-28k and parvalbumin in the rat nervous system. Neurosci. 35:375-475 (1990).
5. Condé F, et al. Local circuit neurons immunoreactive for calretinin, calbindin D‐28k or parvalbumin in monkey prefronatal cortex: Distribution and morphology. J. Comp. Neurol. 341:95-116 (1994).
6. Hof PR, et al. Cellular distribution of the calcium-binding proteins parvalbumin, calbindin, and calretinin in the neocortex of mammals: phylogenetic and developmental patterns. J. Chem. Neuroanat. 16:77-116 (1999).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com