| Name: | Chicken Polyclonal Antibody to Lamin A/C |
| Immunogen: | Full length recombinant human lamin A protein expressed in and purified from E. coli. |
| HGNC Name: | LMNA |
| UniProt: | P02545 |
| Molecular Weight: | 65kDa and 74kDa |
| Host: | Chicken |
| Isotype: | |
| Species Cross-Reactivity: | Human, Rat, Mouse, Horse, Monkey, Dog |
| RRID: | AB_2572338 |
| Format: | Concentrated IgY preparation plus 0.02% NaN3 |
| Applications: | WB, IF/ICC |
| Recommended Dilutions: | WB: 1:2,000. IF/ICC 1:1,000. IHC: 1:2,000 |
| Storage: | Stable at 4°C for one year. |
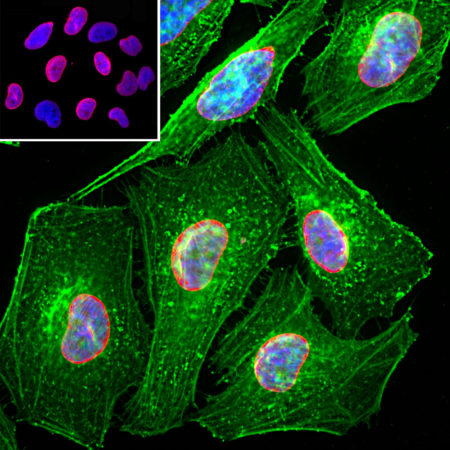
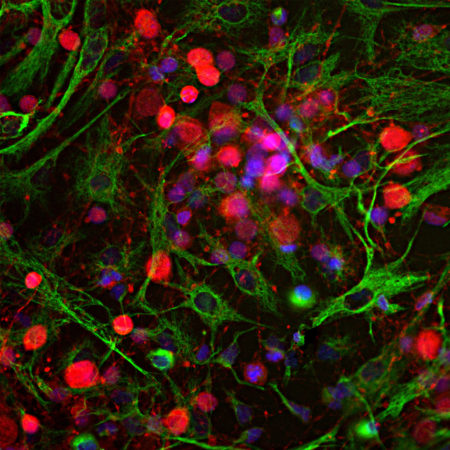
Immunofluorescent analysis of HeLa cells stained with chicken pAb to lamin A/C, CPCA-LaminAC, dilution 1:2,000 in red, and costained with mouse mAb to actin, MCA-5J11, dilution 1:500, in green. The blue is Hoechst staining of nuclear DNA. The CPCA-LaminAC antibody specifically labels the nuclear lamina, while the actin antibody stains the submembranous actin-rich cytoskeleton, stress fibers and bundles of actin associated with cell adhesion sites.
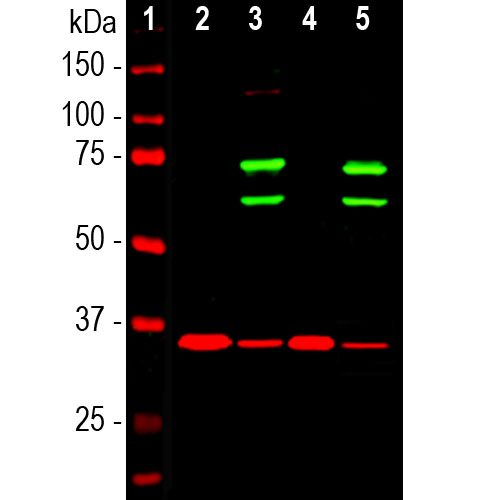
Western blot analysis of cytosolic or nuclear enriched fractions of cell lines probed with chicken pAb to lamin A/C, CPCA-LaminAC, dilution 1:1,000 in green: [1] protein standard (red), [2] HeLa cytosol, [3] HeLa nuclear, [4] NIH-3T3 cytosol, and [5] NIH-3T3 nuclear fractions. Two strong bands at 65kDa and 74kDa correspond to lamin A and lamin C proteins respectively, detected exclusively in the nuclear fractions. The same blot was simultaneously probed with mouse mAb to GAPDH, MCA-1D4, in red. The single band at 37kDa represents GAPDH protein which is expressed predominantly in the cytosolic fractions.
Chicken Polyclonal Antibody to Lamin A/C
Cat# CPCA-LaminAC
$120.00 – $800.00
Lamin A and lamin C are members of the intermediate filament protein family and are located in the nucleus where they function as skeletal components of the inner nuclear membrane (1). The two proteins are generated by alternate transcription from the single LMNA gene. Lamin A has a molecular weight of about 74kDa while lamin C is 65kDa. The lamin A protein includes a C-terminal segment of 98 amino acids missing from lamin C, while lamin C has a unique C-terminal 6 amino acid peptide not present in lamin A. As a result antibodies raised against lamin A are almost certain bind to lamin C. During cell division the nuclear lamina breaks down and lamin A/C containing filaments depolymerize, this being regulated by phosphorylation by cyclin dependent protein kinase 1. Mutations in the lamin A/C gene are associated with several serious human diseases, including Emery-Dreifuss muscular dystrophy, familial partial lipodystrophy, limb girdle muscular dystrophy, dilated cardiomyopathy, Charcot-Marie-Tooth disease type 2B1, Hutchinson-Gilford progeria syndrome and Hutchinson-Gilford progeria syndrome (3-6).
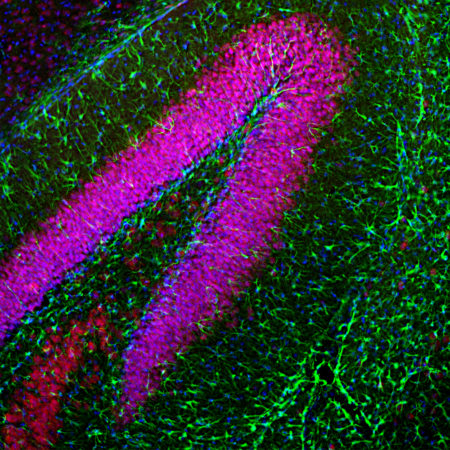
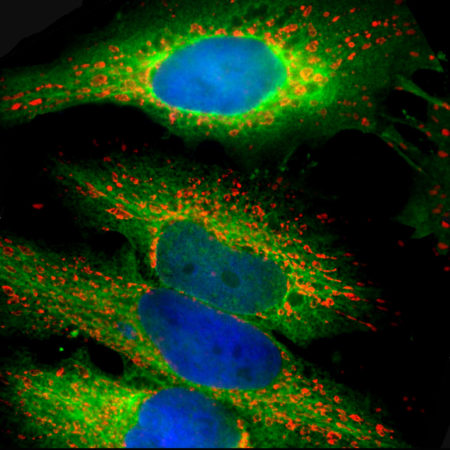
The CPCA-LaminAC was raised against full length recombinant human lamin A, binds human lamin C, and works on western blots with the rodent protein also. It can be used to visualize the nuclear lamina by IF and ICC and also works in IHC, for later data select “Additional Info” tab. We also market a mouse monoclonal antibody to lamin A/C MCA-4C4. Mouse select image above left for larger view.
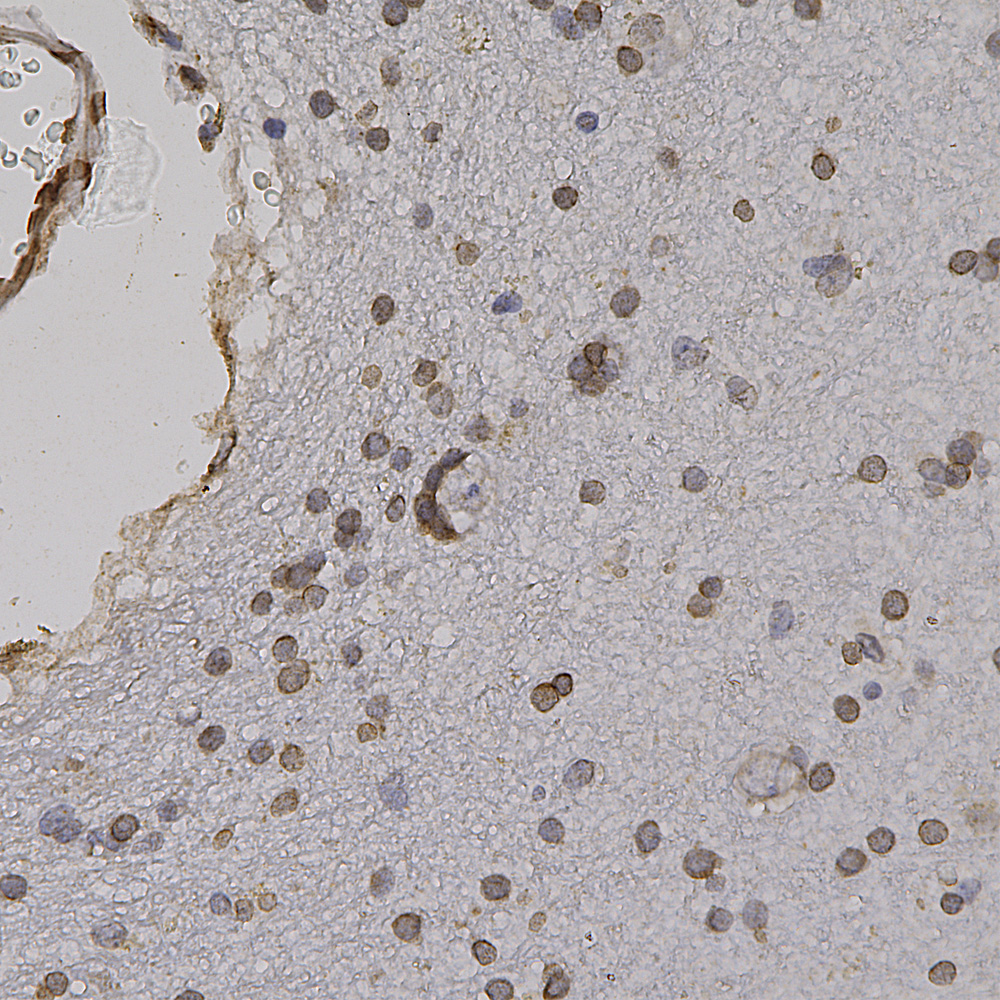
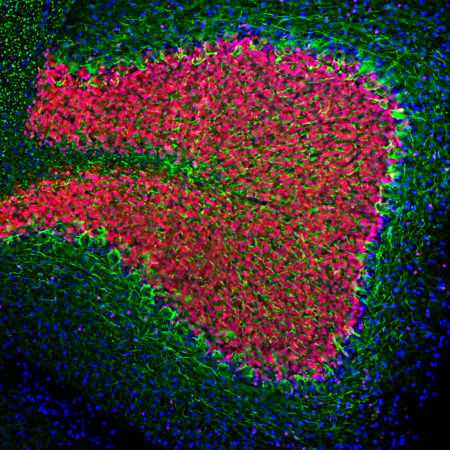
Chromogenic Immunostaining of a formalin fixed paraffin embedded human cerebral cortex section with chicken pAb to lamin A/C, CPCA-LaminAC, dilution 1:2,000, detected in DAB (brown) following the ABC method. Hematoxylin (blue) was used as the counterstain. Lamin A/C antibody recognizes the nuclear lamina, resulting in a distinct ring pattern. Mouse select image at left for larger view.
1. Fisher DZ, Chaudhary N, Blobel G. cDNA sequencing of nuclear lamins A and C reveals primary and secondary structural homology to intermediate filament proteins. PNAS 83:6450-54 (1986).
2. McKeon FD, Kirschner MW, Caput D. Homologies in both primary and secondary structure between nuclear envelope and intermediate filament proteins. Nature 319: 463-8 (1986).
3. Bonne G, et al. Mutations in the gene encoding lamin A/C cause autosomal dominant Emery-Dreifuss muscular dystrophy. Nat. Genet. 21:285-8 (1999).
4. Novelli G, et al. Mandibuloacral dysplasia is caused by a mutation in LMNA-encoding lamin A/C. Am. J. Hum. Genet. 71:426-31 (2002).
5. De Sandre-Giovannoli A, et al. Homozygous Defects In Lmna, Encoding Lamin A/C Nuclear‐Envelope Proteins, Cause Autosomal Recessive Axonal Neuropathy In Human (Charcot‐Marie‐Tooth Disorder Type 2) And Mouse Am. J. Hum. Genet. 70:726-36 (2002).
6. Liu B and Zhou Z. Lamin A/C, laminopathies and premature ageing. Histol. Histopathol. 23:747-63 (2006).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com