| Name: | Mouse Monoclonal Antibody to Aurora A kinase |
| Immunogen: | Full length recombinant human aurora A expressed in and purified from E. coli |
| HGNC Name: | AURKA |
| UniProt: | O14965 |
| Molecular Weight: | 46kDa |
| Host: | Mouse |
| Isotype: | IgG1 |
| Species Cross-Reactivity: | Human, Mouse |
| RRID: | AB_2572228 |
| Format: | Concentrated hybridoma cell culture media plus 5mM NaN3 |
| Applications: | WB, IF/IHC, IHC |
| Recommended Dilutions: | WB: 1:100-1:500. ICC/IF and IHC: 1:100-1:500. |
| Storage: | Store at 4°C for short term, for longer term at -20°C. Avoid freeze/thaw cycles. |
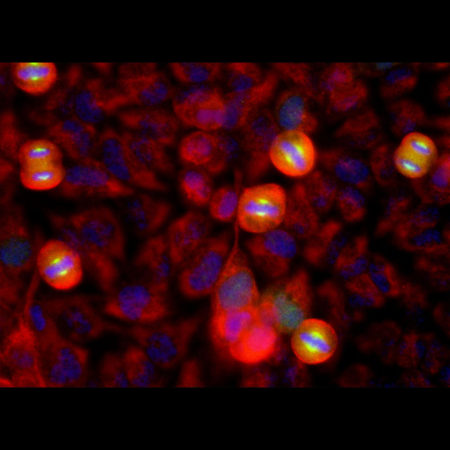
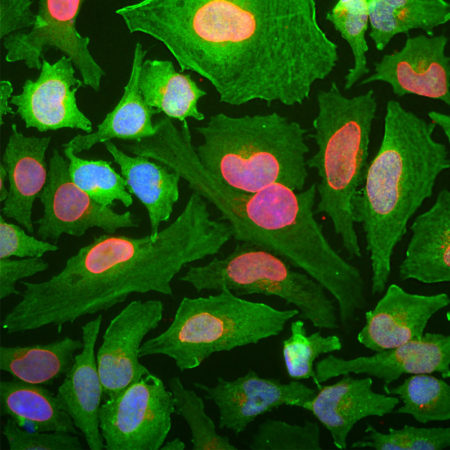
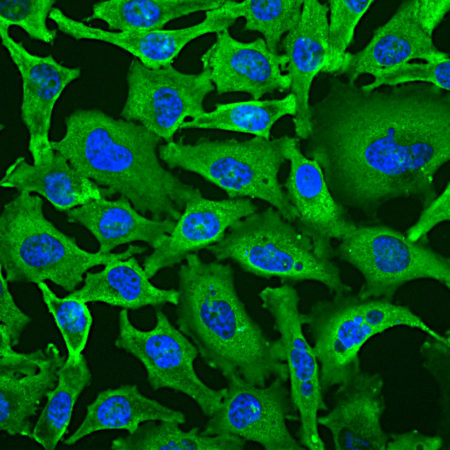
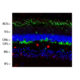
Immunofluorescent analysis of HeLa cell cultures stained with mouse mAb to aurora A kinase, MCA-1A11, in green, and costained with chicken pAb to vimentin, CPCA-Vim, in red. The blue is DAPI staining of nuclear DNA. MCA-1A11 antibody labels aurora A protein expressed in spindle poles at late mitosis.
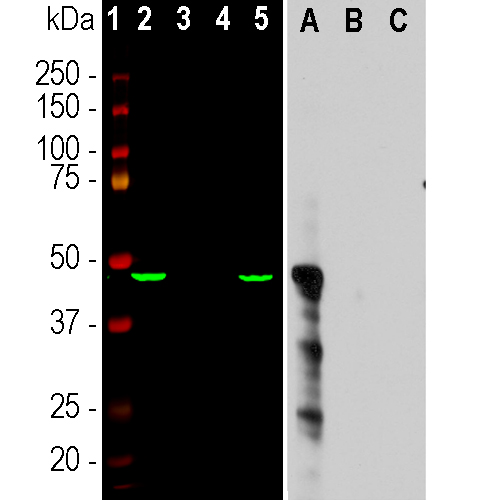
Western blot analysis of different cell lysates and recombinant protein solutions using mouse mAb to aurora A, MCA-1A11. Left: cells were treated with 100ng/mL of nocodazol (a microtubule depolymerizing agent which induces cells to halt at G2/M phase) for 6 hours: [1] protein standard, [2] HeLa, [3] canine A72 cells, [4] Equine NBL6 cells, and [5] murine KR158 cells. The antibody apparently does not recognize canine or equine aurora A, but does bind human and murine. Right: equal amounts of purified human recombinant aurora A, B, C as indicated. The MCA-1A11 antibody reacts with the aurora A recombinant protein but not aurora B or C.
Mouse Monoclonal Antibody to Aurora A kinase
Cat# MCA-1A11
$120.00 – $800.00
Aurora proteins are a family of serine/threonine protein kinases which play a key role in the regulation of cell division which were originally discovered in studies of Drosophila (1). Mammalian genomes encode 3 aurora kinases named aurora A, B and C, each containing a variable regulatory domain at the N terminus followed by a catalytic serine/threonine kinase domain which is almost identical between them, see here for sequence alignment. As a result it is possible to generate antibodies which react with only one aurora kinase or cross react with two or more other kinases. Aurora A and B are almost ubiquitous in distribution while C is normally only expressed in testis. Aurora A is required for centrosome duplication, entry into mitosis, formation of bipolar spindle and mitotic checkpoint (3). Aurora B is a chromosomal passenger protein and essential for chromosome condensation, kinetochore functions, spindle checkpoint activation and cytokinesis completion (4). Aurora C is normally involved in spermatogenesis, but may also be expressed in many transformed cell lines and tumors and has been less well studied to date (5). The aurora kinases are essential for the progression to cell division and as a result there has been much interest in the development of drugs aimed at inhibiting their activity for use as anticancer agents (6,7). We have made a panel of antibodies to the aurora kinases, concentrating originally on aurora A and B, and we made recombinant full length human aurora constructs of all three to document their potential cross reactivity.
MCA-1A11 was made against recombinant human aurora A, and was shown to be non-reactive with aurora B or C. As expected the antibody localizes aurora A in spindle poles and mitotic spindles at late mitosis and recognizes the appropriate sized protein in human and mouse cell lysates on western blots. We also supply other aurora specific antibodies, to both aurora A and B, MCA-5A12 and MCA-3H1, aurora B specific, MCA-6G2, and MCA-3F11. Mouse select image at left for larger view.
1. Glover DM, Leibowitz MH, McLean DA, Parry H. Mutations in aurora prevent centrosome separation leading to the formation of monopolar spindles. Cell 81:95-105 (1995).
2. Hochegger H, Hegarat N, Pereira-Leal JB. Aurora at the pole and equator: overlapping functions of Aurora kinases in the mitotic spindle. Open Biol. 20:120-85 (2013).
3. Barr AR, Gergely F. Aurora-A: the maker and breaker of spindle poles. J. Cell Sci. 120:2987-96 (2007).
4. Andrew PD, Knatko E, Moore WJ, Swedlow JR. Mitotic mechanics: the auroras come into view. Curr. Opin. Cell Biol. 15:672-83 (2003).
5. Tang CJ, Lin CY, Tang TK. Dynamic localization and functional implications of Aurora-C kinase during male mouse meiosis. Dev. Biol. 290:398-410 (2006).
6. Dutertre S, et al. The absence of p53 aggravates polyploidy and centrosome number abnormality induced by Aurora-C overexpression. Cell Cycle 4:1783-7 (2005).
7. Kimura M, Matsuda Y, Yoshioka T, Okano Y. Cell cycle-dependent expression and centrosome localization of a third human aurora/Ipl1-related protein kinase, AIK3. J. Biol. Chem. 274:7334-40 (1999).
8. Takitoh T, et al. Activation of Aurora-A is essential for neuronal migration via modulation of microtubule organization. J. Neurosci. 32:11050-66 (2012).
9. Marumoto T, et al. Aurora-A kinase maintains the fidelity of early and late mitotic events in HeLa cells. J. Biol. Chem. 278:5178-95 (2003).
A sequence alignment of the 3 human aurora molecules can be downloaded from https://encorbio.com/Alignments/Aurora_alignment.pdf.
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com