| Name: | Mouse Monoclonal Antibody to Aldolase C: N-terminus specific |
| Immunogen: | N-terminal sequence MPHSYPALSAEQKKELSDIA |
| HGNC Name: | ALDOC |
| UniProt: | P09972 |
| Molecular Weight: | 40kDa |
| Host: | Mouse |
| Isotype: | IgG1 heavy, κ light |
| Species Cross-Reactivity: | Human, rat, mouse, cow, pig |
| RRID: | AB_2571880 |
| Format: | Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IIF/ICC, IHC |
| Recommended Dilutions: | WB: 1:2,000. IF/ICC or IHC: 1:1,000. |
| Storage: | Store at 4°C for short term, for longer term at -20°C. Avoid freeze / thaw cycles. |

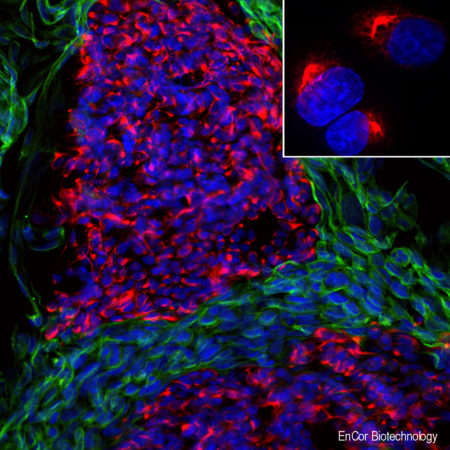
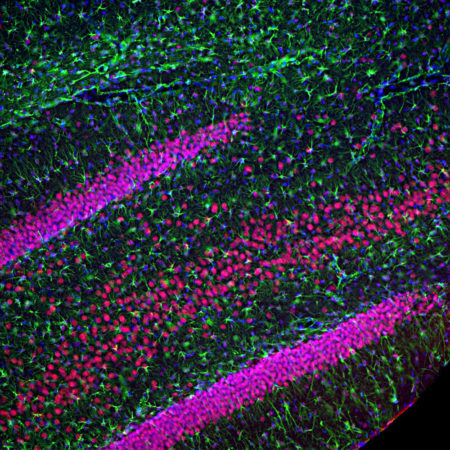
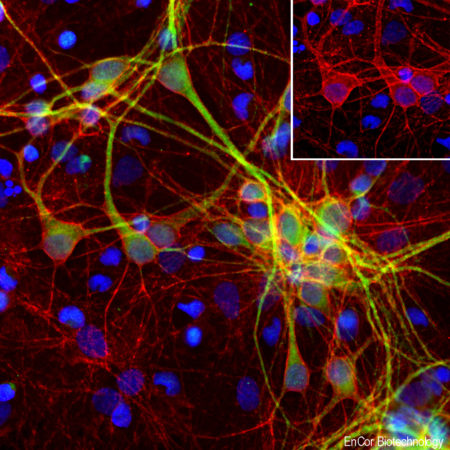
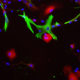
A. Immunofluorescence of rat cerebellum section stained with mouse mAb to aldolase C, MCA-4A9, dilution 1:1,000, in green, and costained with rabbit pAb to GFAP, RPCA-GFAP, dilution 1:5,000 in red. The blue is Hoechst staining of nuclear DNA. Aldolase C antibody selectively labels the perikarya and dendrites of Purkinje cells, while GFAP antibody stains processes of Bergman glia and astrocytic cells. B. Immunofluorescence of E20 rat cortical culture stained with MCA-4A9, dilution 1:1,000 in green, and costained with chicken pAb to MAP2, CPCA-MAP2, dilution 1:10,000 in red. The blue is Hoechst staining of nuclear DNA. Aldolase C antibody labels cytosolic protein expressed in glial cells, while MAP2 antibody stains dendrites and perikarya of mature neurons.
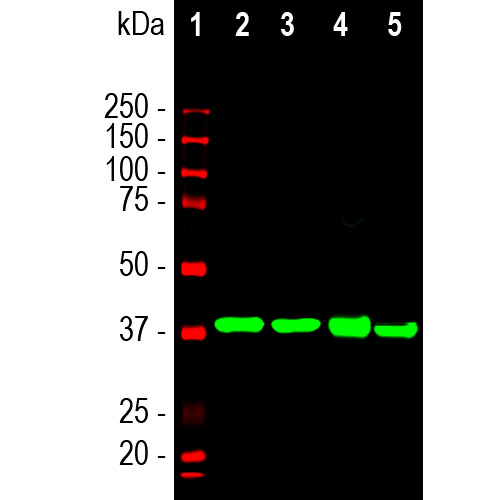
Western blot analysis of different tissue lysates using mouse mAb to aldolase C, MCA-4A9, dilution 1:2,000 in green: [1] protein standard (in red), [2] rat brain, [3] mouse brain, [4] cow cerebellum, and [5] pig hippocampus. The single strong band at about 40kDa corresponds to Aldolase C protein.
Mouse Monoclonal Antibody to Aldolase C
Cat# MCA-4A9
$120.00 – $800.00
Aldolases are important glycolytic cytosolic enzymes which catalyse the reversible conversion of fructose-1,6-bisphosphate to glyceraldehyde 3-phosphate. There are three aldolase isozymes coded by three distinct genes in mammals, namely aldolases A, B, and C. Aldolase A is heavily expressed in muscle, and aldolase B is a liver-specific enzyme (1-5). In the adult, aldolase C is the brain-specific isozyme expressed in astrocytes and a few classes of neurons, notably Purkinje cells (4). Appropriate antibodies to aldolase C are therefore useful to identify astrocytes in cell culture and sections, and the enzyme may be over expressed in some forms of cancer (6). Recent studies also suggest that detection of aldolase C in blood may be a useful marker of the severity of traumatic brain injury (7).
The MCA-4A9 antibody was made against a recombinant construct including the N-terminal peptide of human aldolase C. Later mapping studies localized the epitope to the peptide HSYPALSAEQKKELSDIA, amino acids 3-20. Since the three aldolase enzymes are quite similar in amino acid sequence many available antibodies to one protein have usually undocumented cross-reactivity with the other two. However we have used appropriate recombinant constructs to shown that MCA-4A9 is completely specific for aldolase C with no reactivity with either aldolase A or B. Since we know the identity of the peptide used to generate MCA-4A9 is firmly mapped to the N-terminus of aldolase C, a region in which there is considerable variability between the three aldolase enzymes. This antibody works well on western blots and for IF, ICC and IHC. Mouse select image at left for larger view.
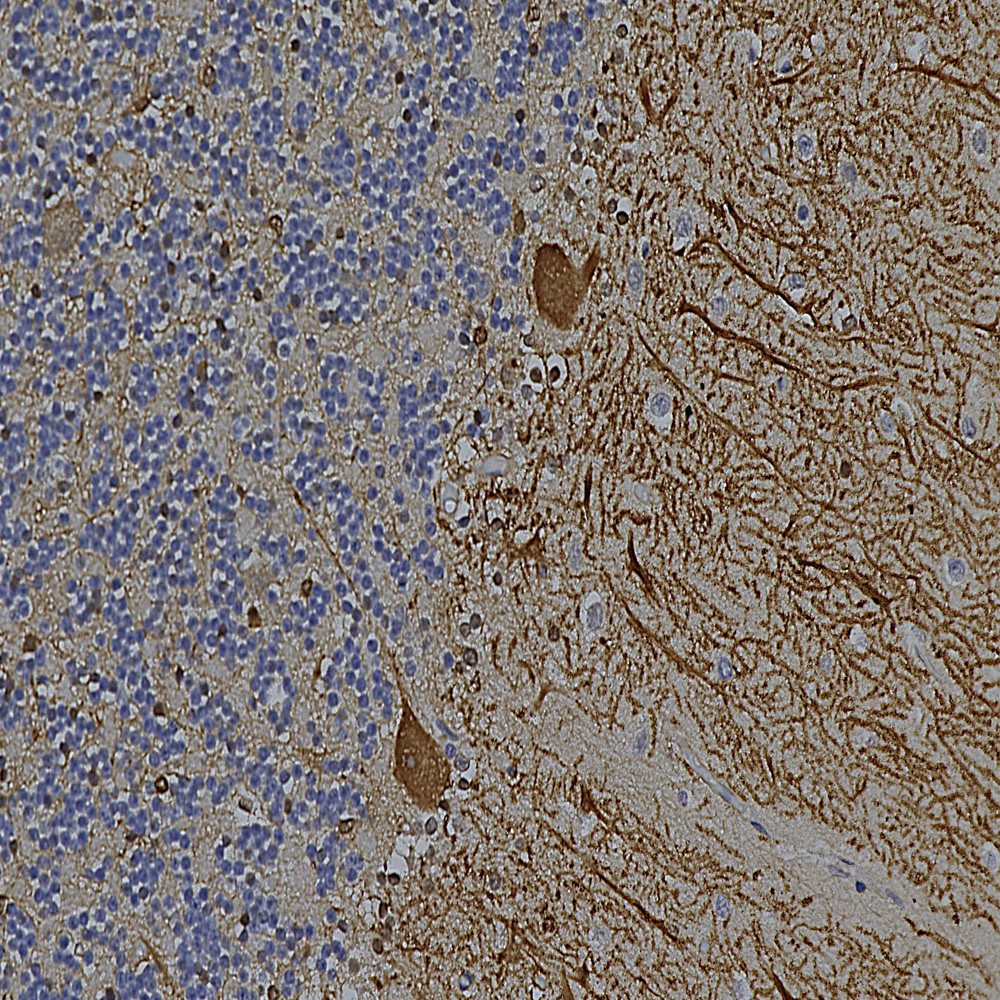
Chromogenic immunostaining of a NBF fixed paraffin embedded human cerebellum section with mouse mAb to Aldolase C, MCA-4A9, dilution 1:2,000, detected with DAB (brown) using the Vector Labs ImmPRESS method and reagents with citra buffer retrieval. Hematoxylin (blue) was used as the counterstain. The MCA-4A9 antibody strongly labels the cell bodies and dendrites of Purkinje cells. This antibody performs well in testing with both 4% PFA and standard NBF fixed tissues.Mouse select image for larger view.
Immunofluorescent analysis of cortical neuron-glial cell culture from E20 rat stained with mouse mAb to aldolaseC, MCA-4A9, dilution 1:1,000 in green, and costained with chicken pAb to MAP2, CPCA-MAP2, dilution 1:10,000 in red. The blue is Hoechst staining of nuclear DNA. The aldolase C antibody labels cytosolic protein expressed in the glial cells, while theMAP2 antibody stains dendrites and perikarya of mature neurons.
1. Popovici T, et al. Localization of aldolase C mRNA in brain cells. FEBS Lett. 268:189-193 (1990).
2. Weber A, et al. Dietary Control of Aldolase B and L-type Pyruvate Kinase RNAs in Rat. J. Biol. Chem. 259:1798-802 (1984).
3. Mukai T, et al. The structure of the brain-specific rat aldolase C gene and its regional expression. BBRC 174:1035-42 (1991).
4. Royds J, et al. Monoclonal antibody to aldolase C: a selective marker for Purkinje cells in the human cerebellum. Neuropathol. Appl. Neurobiol. 13:11-21 (1987).
5. Thompson R., Kynoch P. Willson V. Cellular localization of aldolase C subunits in human brain. Brain Res. 232:489-93 (1982).
6. Schapira F, Reuber M, Hatzfeld A. Resurgence of two fetal-type of aldolases (A and C) in some fast-growing hepatomas. BBRC 40:321-27 (1970).
7. Halford J, et al. New astroglial injury-defined biomarkers for neurotrauma assessment. J. Cereb. Blood Flow Metab. 37:3278-99 (2017).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com