| Name: | Mouse monoclonal antibody to arrestin 1/S-antigen |
| Immunogen: | Recombinant bovine arrestin 1 with the first 20 amino acids of the C-terminus truncated |
| HGNC Name: | SAG |
| UniProt: | P10523 |
| Molecular Weight: | 48kDa |
| Host: | Mouse |
| Isotype: | IgG1 |
| Species Cross-Reactivity: | Human, rat, mouse, cow, pig, horse |
| RRID: | AB_2572227 |
| Format: | Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:5,000 IF/ICC: 1:1,000. IHC: 1:1,000. |
| Storage: | Stable at 4°C for one year, for longer term store at -20°C |
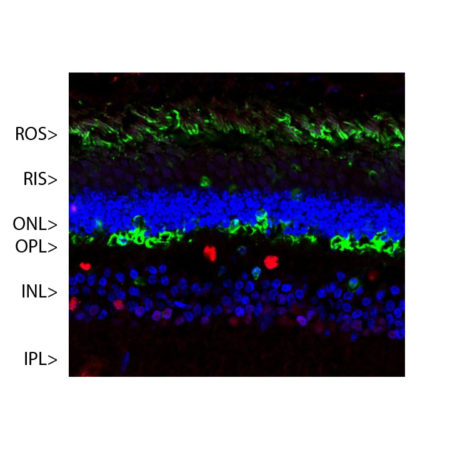
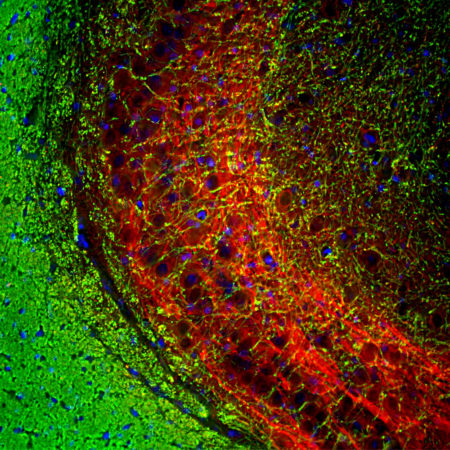
Confocal image of a pig retina stained with MCA-S128 (green). Arrestin 1 is most abundant in the outer segments (ROS) and inner surface of the outer nuclear layer (ONL), and can be used to identify components of rod photoreceptor cells. (Cone photoreceptors have a different arrestin isotype). Other retinal layers are inner segments (IS), outer plexiform layer (OPL), inner nuclear layer (INL) and inner plexiform layer (IPL). The red stain shows staining for FOX2, an RNA binding nuclear protein related to Fox3/NeuN, which stains nuclei of horizontal neurons and some other neurons in the INL and IPL. Nuclear DNA was revealed with DAPI (blue).
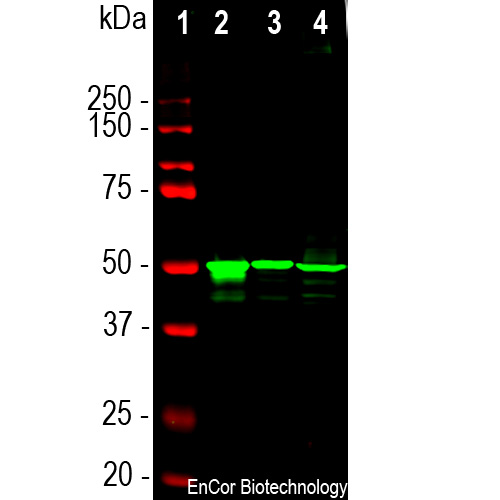
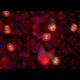
Western blot analysis of retina lysates from different species using mouse mAb to arrestin 1, MCA-S128, dilution 1:5,000 in green: [1] protein standard (red), [2] rat [3] mouse and [4] cow retina lysates. The MCA-S128 antibody detects arrestin 1 a.k.a. visual arrestin and S-antigen, running at about 48kDa.
Mouse Monoclonal Antibody to Arrestin1
Cat# MCA-S128
$120.00 – $800.00
The arrestin proteins are a family of regulators of G protein-coupled receptor (GPCR) signaling. The retina contains one family member, visual arrestin or arrestin 1, which is localized in the rod outer segments where it binds to and regulates the activity of phosphorylated rhodopsin. Other arrestin family members are cone-arrestin, β-arrestin 1 (a.k.a. arrestin 2) and β-arrestin 2. The arrestin 1 protein is also known as S-antigen, as it was originally identified since autoantibodies to this protein are seen in experimental allergic uveitis, an animal model of human uveitis (1). A series of further studies revealed details of how the protein functioned in the retina (2-4). Arrestin family proteins bind to activated and phosphorylated G-protein receptors, “arresting” their G-protein mediated signaling (5). Recent evidence suggests that arrestin family members have a wide variety of other functions, such as mediating endocytosis of certain G-protein coupled receptors and activating Src and MAP kinase signaling among other functions (6). Arrestin proteins therefore appear to function as versatile multipurpose signaling adapters.
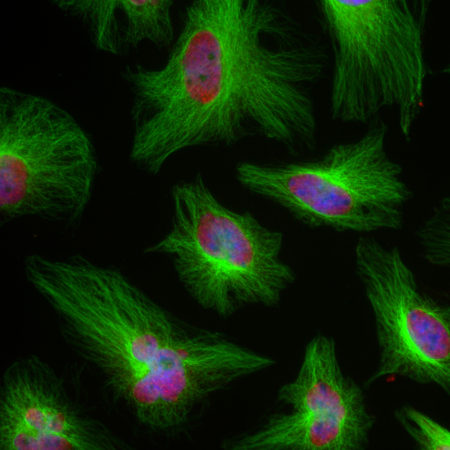
The MCA-S128 antibody was made against recombinant bovine arrestin 1 with 20 N-terminal amino acids truncated (7). The antibody works well for western blotting and for IF, ICC and IHC (for IHC see data under “Additional Info” tab). The antibody does not cross-react with cone-arrestin, β-arrestin 1 or β-arrestin 2. The MCA-S128 antibody can be used to study this protein in cells, tissues and tissue lysates. Mouse select image above left for larger view.
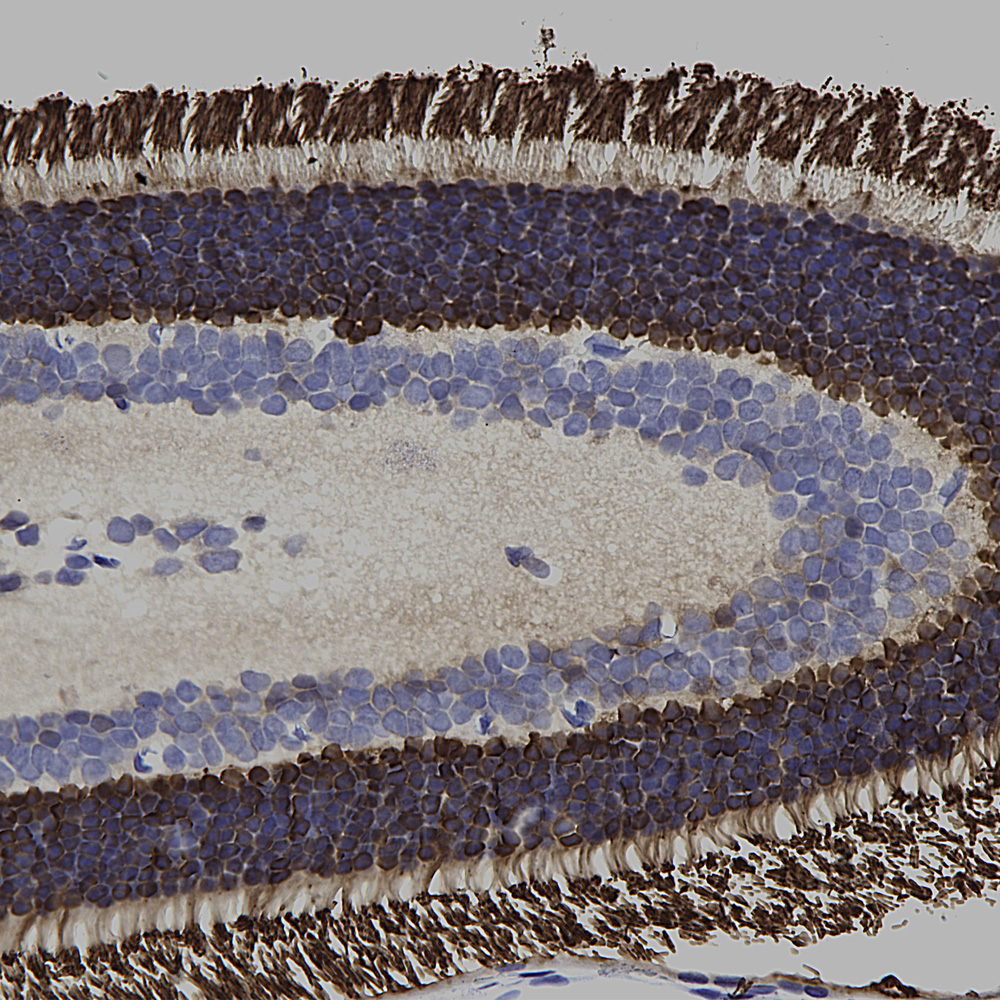
Chromogenic immunostaining of a 4% PFA fixed paraffin embedded rat retina section with mouse mAb to visual arrestin 1, MCA-S128, dilution 1:1,000, detected in DAB (brown) following the ImmPress method. Hematoxylin (blue) was used as the counterstain. In this image, MCA-S128 strongly labeled the inner surface of the outer nuclear layer of the retina. This antibody has not been tested in NBF fixed material.
1. Wacker WB, et al. Experimental allergic uveitis. Isolation, characterization, and localization of a soluble uveitopathogenic antigen from bovine retina. J. Immunol. 119:1949-58 (1977).
2. Kühn H, Hall SW, Wilden U. Light-induced binding of 48-kDa protein to photoreceptor membranes is highly enhanced by phosphorylation of rhodopsin. FEBS Lett. 176:473-8 (1984).
3. Pfister C, et al. Retinal S antigen identified as the 48K protein regulating light-dependent phosphodiesterase in rods. Science 228:891-3 (1985).
4. Wilden U, Hall SW, Kühn H. Phosphodiesterase activation by photoexcited rhodopsin is quenched when rhodopsin is phosphorylated and binds the intrinsic 48-kDa protein of rod outer segments. PNAS 83:1174-8 (1986).
5. Lohse MJ, et al. β-arrestin: a protein that regulates beta-adrenergic receptor function. Science 248:1547-50 (1990).
6. Peterson YK, Luttrell LM. The Diverse Roles of Arrestin Scaffolds in G Protein–Coupled Receptor Signaling.
Pharmacol. Rev. 69:256–297 (2017).
7. Smith WC, et al. Identification of regions of arrestin that bind to rhodopsin. Biochem. 38:2752-61 (1999).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com