| Name: | Rabbit polyclonal antibody to adenylate cyclase III |
| Immunogen: | C-terminal peptide of rat ACIII, PAAFPNGSSVTLPHQVVDNP with a Cys added to the N-terminus to allow coupling to KLH. |
| HGNC Name: | ADCY3 |
| UniProt: | P21932 |
| Molecular Weight: | 130-200kDa by SDS-PAGE |
| Host: | Rabbit |
| Isotype: | |
| Species Cross-Reactivity: | Human, rat, mouse |
| RRID: | AB_2572219 |
| Format: | Affinity Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:1,000. IF/ICC or IHC: 1:10,000-20,000. |
| Storage: | Store at 4°C for short term, for longer term store at -20°C |
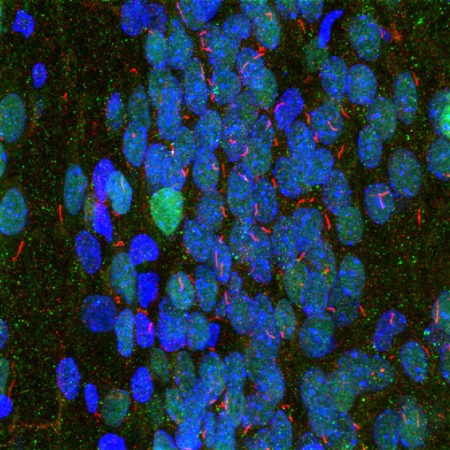
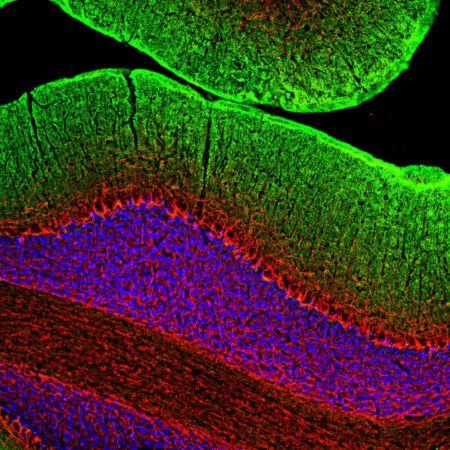
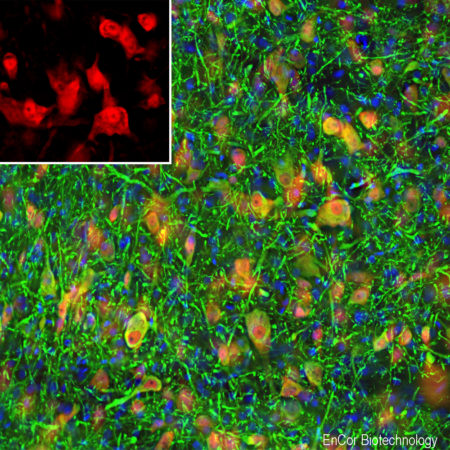
Immunofluorescence analysis of rat hippocampus section stained with rabbit pAb to to adenylate cyclase III, RPCA-ACIII, in red and costained with mouse mAb to MECP2, MCA-5H12, in green. The blue is DAPI staining of nuclear DNA. The ACIII antibody reveals neuronal cilia while the MECP2 antibody reveals the nuclei of certain neurons to a variable degree. This is a stacked confocal image made with a 100X objective lens.
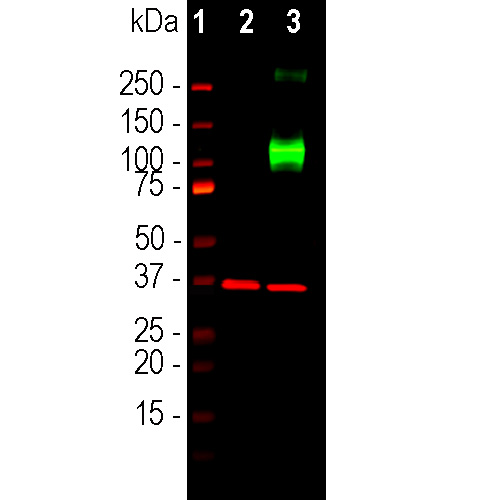
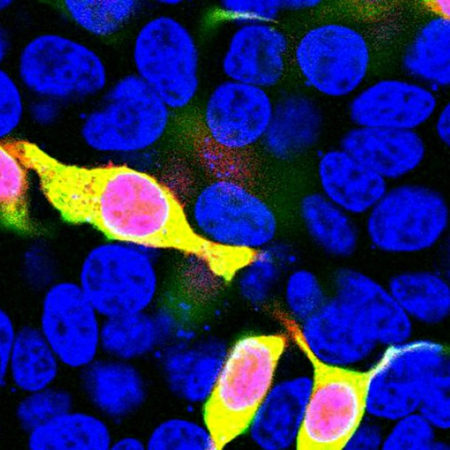
Western blot analysis of HEK293 cell lysates using rabbit pAb to ACIII, RPCA-ACIII, dilution 1:2,000, in green: [1] protein standard, [7] non-transfected HEK293 cells, and [8] HEK293 cells transfected with an expression construct containing a Myc-DDK tagged full length human adenylate cyclase III cDNA. The strong band at about 130kDa in the transfected cells demonstrates overexpression of the human ACIII protein, and the bands over 250kDa presumably correspond to either glycosylated or aggregated forms of ACIII. The same blot was simultaneously probed with mouse mAb to GAPDH, MCA-1D4, dilution 1:5,000, in red, which reveals the single band at ~37kDa in both transfected and non-transfected cells.
Rabbit Polyclonal Antibody to Adenylate Cyclase III
Cat# RPCA-ACIII
$150.00 – $1,000.00
G-protein coupled receptors are a large and variable family of membrane proteins. On binding their specific ligand they activate specific members of the family of trimeric G-proteins which in turn activate other signaling enzymes. Adenylate cyclases are one of these and are activated by the GTP bound GαS subunits of trimeric G-proteins. Adenylate cyclases are responsible for the production of the important “second messenger” signaling molecule cyclic-AMP which in turn activates the cAMP dependent protein kinase. This kinase phosphorylates numerous substrate molecules on serine or threonine residues and so alters their activity. There are several different adenylate cyclase genes and protein products with each have distinctly different distribution patterns in cells and tissues. The type III adenylate cyclase enzyme is specifically localized in the membranes surrounding neuronal cilia, and is activated by specific G-protein coupled receptors also located in cilia (1-5). Neuronal cilia express a variety of other receptors types and mediators of other signaling pathways and appear to function as a unique and complex neuronal sensory structure (1-5). For examples, the somatostatin 3 receptor, neuropeptide Y 2 receptor and melanin concentrating hormone receptor 1 are localized in neuronal cilia and the sonic hedgehog and Wnt signaling pathway act on neurons primarily through neuronal cilia (6).
The RPCA-ACIII antibody was made against the extreme C-terminal peptide of rat ACIII, PAAFPNGSSVTLPHQVVDNP, amino acids 1125-1144 of the Genbank entry NP_570135.2. A cysteine residue was added to the N-terminus to allow coupling to MBS-activated keyhole limpet hemocyanin. The antibody works on mouse cells which express the same peptide and also on human cells, presumably because the corresponding peptide in the human AC3 sequence is the closely related peptide LATFPNGPSVTLPHQVVDNS. In line with this the antibody binds full length transfected human ACIII on western blots. The antibody works well to identify neuronal cilia on human and rodent cells. We have also generated a mouse monoclonal and a chicken polyclonal antibody to the same ACIII peptide, MCA-1A12 and CPCA-ACIII. Mouse select image at left for larger view.
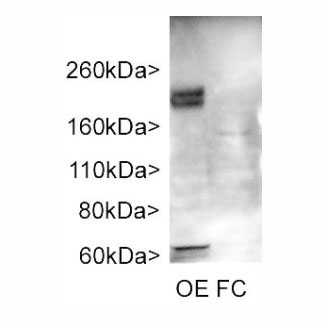
Western blots of rat olfactory epithelium (OE) and frontal cortex (FC). Our antibody stains bands at about 200kDa in olfactory epithelium, a tissue which is rich in cilia. Fewer cilia are found in frontal cortex, and the protein is also less heavily glycosylated, so that a much less prominent band is seen at about 160kDa. Data generated in the laboratory of Matt Sarkisian in the University of Florida.
This antibody has been cited in peer reviewed literature, see the CiteAb link here.
1. Fuchs JL, Schwark HD. Neuronal primary cilia: a review. Cell Biol. Int. 28:111-8 (2004).
2. Louvi A and Grove EA. Cilia in the CNS: the quiet organelle claims center stage. Neuron 69:1046-60 (2011).
3. Singla V, Reiter JF. The primary cilium as the cell’s antenna: signaling at a sensory organelle. Science 313:629-33 (2006).
4. Green JA, Mykytyn K. Neuronal Primary Cilia: An Underappreciated Signaling and Sensory Organelle in the Brain. Neuropsychopharm. 39:244–5 (2014).
5. May-Simera HL, Kelley MW. Cilia, Wnt signaling, and the cytoskeleton. Cilia 2;1:7 (2012).
6. Guemez-Gamboa A, et al. Primary cilia in the developing and mature brain. Neuron 82:511-21 (2014).
7. Guadiana SM, et al. Arborization of Dendrites by developing neocortical neurons is dependent on primary cilia and Type 3 adenylyl cyclase. J. Neurosci. 33:2626-38 (2013).
This antibody has been cited in peer reviewed literature, see the CiteAb link here.
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com