| Name: | Rabbit polyclonal antibody to S. aureus CAS9 |
| Immunogen: | CAS9 from S. aureus expressed in and purified from E. coli |
| HGNC Name: | N.A. |
| UniProt: | J7RUA5 |
| Molecular Weight: | 124kDa |
| Host: | Rabbit |
| Isotype: | |
| Species Cross-Reactivity: | Staphylococcus aureus |
| RRID: | AB_2744685 |
| Format: | Antibody is supplied as rabbit serum |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:1,000-1:5,000. IF/ICC and IHC:1:5,000 |
| Storage: | Storage for short term at 4°C recommended, for longer term at -20°C, minimize freeze/thaw cycles |
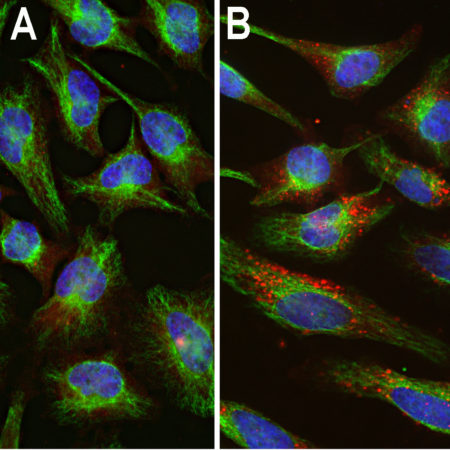
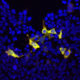
HEK293 cells were transfected with a construct including the C-terminal 250 amino acids of S. aureus CAS9 fused to GFP and stained with RPCA-CAS9-Sa in red. Transfected cells express the green fusion protein and bind the antibody in red, producing a yellow signal. Nuclear DNA in transfected and non-transfected cells is revealed with the blue DNA stain DAPI.
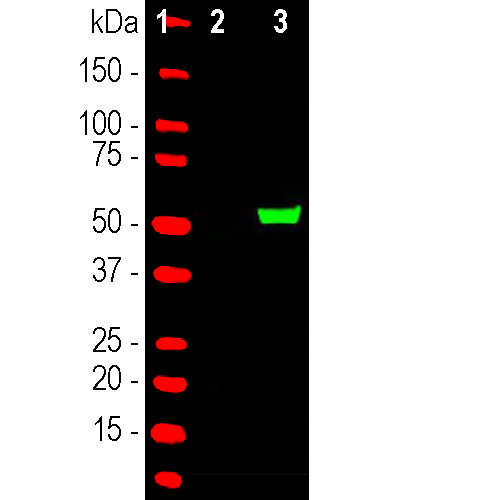
Western blot analysis of HEK293 cell lysates using rabbit pAb to S. aureus CAS9, RPCA-CAS9-Sa: [1] protein standard (red), [2] nontransfected cells and [3] transfected cells with GFP-CAS9 (C-terminal 803-1053 amino acids of S. aureus CAS9) fusion conctruct. The band at about 53kDa corresponds to the GFP-CAS9 fusion protein.
Rabbit Polyclonal Antibody to S. aureus CAS9
Cat# RPCA-CAS9-Sa
$120.00 – $800.00
A recent revolution in biology has been stimulated by the discovery of CRISPR, or “Clustered Regularly Interspaced Short Palindromic Repeats” and the understanding of the “CRISPR Associated” enzymes (CAS 1,2). The CRISPR repeated sequences are found in bacterial genomes and function as part of unique bacterial immune system which contain short DNA sequences derived from viruses which have infected the bacteria. These virally derived sequences can make short RNA sequences which can hybridize with specific viral DNA and target a nuclease, such as CAS9, to the viral sequence. So CAS9 is directed to cleave the specific viral sequence and so inactivate the virus. The RNA sequence can be designed to specifically cut DNA virtually anywhere, including in the genomes of living human and other mammalian cells, allowing inexpensive gene editing with unprecedented ease. For example three groups of researchers essentially cured the disease state in a mouse model of Duchenne muscular dystrophy (3-5). A similar approach essentially cured dogs affected with a related disease state (6). Several varieties of CAS9 have been studied and there are several other related enzymes with similar properties. Much of the early work was performed with CAS9 from Streptococcus pyogenes which is rather large at ~158kDa, so the corresponding DNA is also rather large at about 4.2kb. This is problematic with some expression systems especially since DNA encoding RNA sequences and possibly other regulatory elements are usually required. The CAS9 gene of Staphylococcus aureus is significantly smaller, 3kb, producing a protein of 124kDa (6). For an excellent recent review of the various CAS family enzymes and their utility see reference 8.
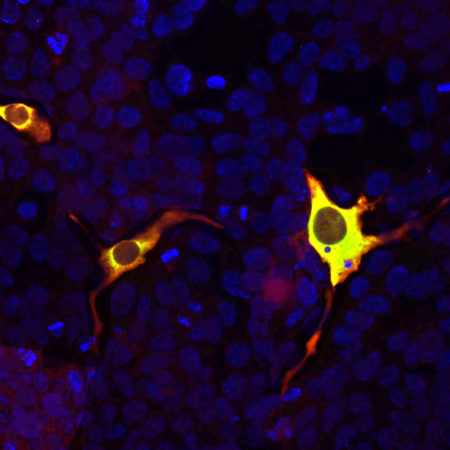
The RPCA-CAS9-Sa antibody can be used to verify the expression of S. aureus CAS9 in cells and in tissues. We used the same immunogen to generate a chicken polyclonal and a mouse monoclonal to S. aureus, CPCA-CAS9-Sa and MCA-6F7. Mouse select image at left for larger view.
The S. pyogenes Cas9 sequence was based on that was UniProt J7RUA5 which was inserted into the eukaryotic expression vector pET30a(+) which adds an N-terminal His-tag and some other sequence, underlined below. This sequence includes a thrombin cleavage site (blue), an S-tag affinity peptide (red) and an enterokinase cleavage site (green).
HHHHHHSSGL VPRGSGMKET AAAKFERQHM DSPDLGTDDD DKAMADIGSE FNNLNGLYDK 60
DNDKLKKLIN KSPEKLLMYH HDPQTYQKLK LIMEQYGDEK NPLYKYYEET GNYLTKYSKK 120
DNGPVIKKIK YYGNKLNAHL DITDDYPNSR NKVVKLSLKP YRFDVYLDNG VYKFVTVKNL 180
DVIKKENYYE VNSKCYEEAK KLKKISNQAE FIASFYNNDL IKINGELYRV IGVNNDLLNR 240
IEVNMIDITY REYLENMNDK RPPRIIKTIA SKTQSIKKYS TDILGNLYEV KSKKHPQIIK 300
KG 302
References and documentation are available.
Number of amino acids: 302
Molecular weight: 35288.20
Theoretical pI: 9.16
Amino acid composition:
Ala (A) 10 3.3%
Arg (R) 9 3.0%
Asn (N) 27 8.9%
Asp (D) 24 7.9%
Cys (C) 1 0.3%
Gln (Q) 7 2.3%
Glu (E) 18 6.0%
Gly (G) 15 5.0%
His (H) 11 3.6%
Ile (I) 22 7.3%
Leu (L) 26 8.6%
Lys (K) 43 14.2%
Met (M) 7 2.3%
Phe (F) 6 2.0%
Pro (P) 11 3.6%
Ser (S) 16 5.3%
Thr (T) 11 3.6%
Trp (W) 0 0.0%
Tyr (Y) 24 7.9%
Val (V) 14 4.6%
Total number of negatively charged residues (Asp + Glu): 42
Total number of positively charged residues (Arg + Lys): 52
Extinction coefficients:
This protein does not contain any Trp residues. Experience shows that
this could result in more than 10% error in the computed extinction coefficient.
Extinction coefficients are in units of M-1 cm-1, at 280 nm measured in water.
Ext. coefficient 35760
Abs 0.1% (=1 g/l) 1.013, assuming all pairs of Cys residues form cystines
Ext. coefficient 35760
Abs 0.1% (=1 g/l) 1.013, assuming all Cys residues are reduced
1. Hsu PD, Lander ES, Zhang F. Development and Applications of CRISPR-Cas9 for Genome Engineering. Cell 157:1262-78 (2014).
2. Doudna1 JA, Charpentier E. The new frontier of genome engineering with CRISPR-Cas9 Science 346:1077-86 (2014)
3. Long C, et al. Postnatal genome editing partially restores dystrophin expression in a mouse model of muscular dystrophy. Science 351:400-3 (2015).
4. Nelson CE, et al. In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy. Science 351:403-7 (2015).
5. Tabebordbar M, et al. In vivo gene editing in dystrophic mouse muscle and muscle stem cells. Science 351:407-11 (2015).
6. Amoasii L. et al. Gene editing restores dystrophin expression in a canine model of Duchenne muscular dystrophy.
Science doi:10.1126/science.aau1549 (2018).
7. Ran FA, et al. In vivo genome editing using Staphylococcus aureus Cas9. Nature 520:186-91 (2015).
8. Knott GJ, Doudna J. CRISPR-Cas guides the future of genetic engineering. Science 361:866-9 (2018).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com