| Name: | Rabbit polyclonal antibody to FOX3/NeuN |
| Immunogen: | Peptide corresponding to amino acids 5-24 of human FOX3 coupled to KLH |
| HGNC Name: | RBFOX3 |
| UniProt: | A6NFN3 |
| Molecular Weight: | 46, 48kDa |
| Host: | Rabbit |
| Isotype: | |
| Species Cross-Reactivity: | Human, rat, mouse |
| RRID: | AB_2572282 |
| Format: | Affinity purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM azide |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:500-1:1000. IF/IHC 1:5,000-1:10,000 |
| Storage: | 4°C for short term and -20°C for longer term |
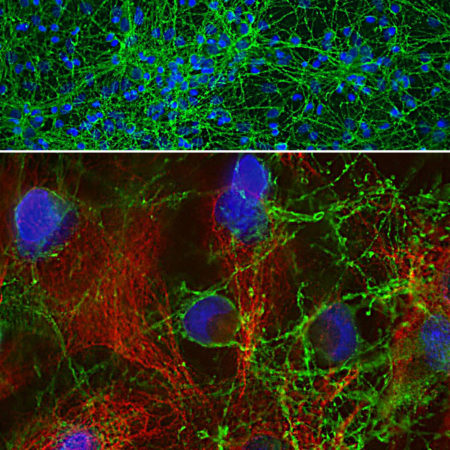
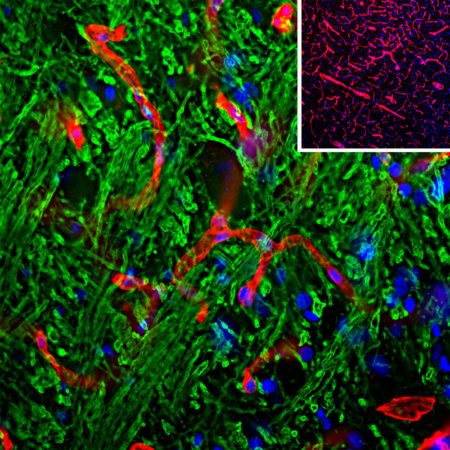
Immunofluorescent analysis of a rat brain section stained with rabbit pAb to FOX3/NeuN protein, RPCA-FOX3, dilution 1:2,000 in red, and costained with chicken pAb to microtubule associated protein 2 (MAP2), CPCA-MAP2, dilution 1:5,000 in green. Following transcardial perfusion of rat with 4% paraformaldehyde, brain was post fixed for 24 hours, cut to 45μm, and free-floating sections were stained with the above antibodies. The FOX3/NeuN antibody selectively stains nuclei and cytoplasm of neuronal cells, while the MAP2 antibody labels dendrites and overlaps with Fox3/NeuN staining in neuronal perikarya.
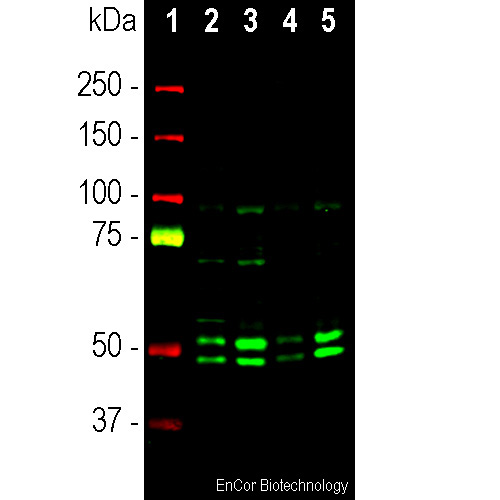
Western blot analysis of cytosolic (cyt) and nuclear enriched (nuc) fractions of whole brain lysates using rabbit pAb to FOX3/NeuN N-terminal peptide, RPCA-FOX3, dilution 1:1,000 in green: [1] protein molecular weight standard (red), [2] rat cyt, [3] rat nuc, [4] mouse cyt, and [5] mouse nuc lysate. Two bands of 46 and 48kDa correspond to the two alternate transcripts of the FOX3/NeuN protein. Western blot was performed under non reducing conditions.
Rabbit Polyclonal Antibody to FOX3/NeuN
Cat# RPCA-FOX3
$150.00 – $1,000.00
In the early 90s an unusual protocol resulted in the raising of a mouse monoclonal antibody against a component of neuronal nuclei and proximal perikarya (1). The component was therefore named “NeuN” and was shown to correspond to two protein bands at 46 and 48kDa in SDS-PAGE blots. The antibody become very widely used as a reliable neuronal marker, apparently binding to neurons in all vertebrates. A few neuronal cell types were not recognized by the original NeuN antibody such as cerebellar Purkinje cells, olfactory mitral cells and many type of retinal neuron. However the vast majority of neurons are strongly NeuN positive, and NeuN immunoreactivity has therefore been widely used to identify neurons.The identity of the NeuN protein was however unknown until 2009 when Kim et al. (2) showed that it was identical to FOX3, a mammalian homolog of a gene product originally identified in Caenorhabditis elegans and named FOX1 (2). The C. elegans protein was discovered as it had a role in sex determination during early development, FOX being an acronym for “feminizing locus on the X chromosome” (3). There are three mammalian FOX1 protein homologs, FOX1, FOX2 and FOX3, which are believed to have a role in the regulation of mRNA splicing (4). All three contain an almost identical central RNA recognition motif or RRM domain, a region of about 90 amino acids found in numerous proteins. The differing protein isoforms of FOX3 result from alternate splicing of two exons which code for an insert close to the C-terminus and a short C-terminal extension (5). The extension includes a C-terminal proline-tyrosine sequence preceded by hydrophobic amino acids (Φ-PY) which is known to target proteins to the nucleus, apparently accounting for FOX3 being present in both nuclei and cytoplasm in certain neurons (5).
The RPCA-FOX3 antibody was raised against an N-terminal sequence amino acids of human FOX3, specifically YPPAQYPPPPQNGIPAEYAP, coupled to keyhole limpet hemocyanin. This amino acids 5-24 of the human sequence. We used this approach since we found that all FOX3 antibodies we tested, both ours and those from other labs, were unable to bind FOX3 in the presence of this peptide, showing that this peptide contains the epitope for several antibodies. The specific data we obtained suggest that the central 10 amino acids, YPPPPQNGIP, are likely the core antigenic region. Since this is the case we decided to make polyclonal antibodies to this peptide and RPCA-FOX3 is one result. We have several other FOX3 antibodies made in chicken, goat, and mouse, CPCA-FOX3, GPCA-FOX3, and MCA-1B7 respectively. All of these antibodies work in the same way as RPCA-FOX3 and the original NeuN antibody and are versatile reagents which can be used in double and triple staining protocols. FOX3/NeuN antibodies can be used to quantify the neuron/glial ratio in primary cell culture and tissue sections of different species (6,7). Mouse select image at left for larger view.
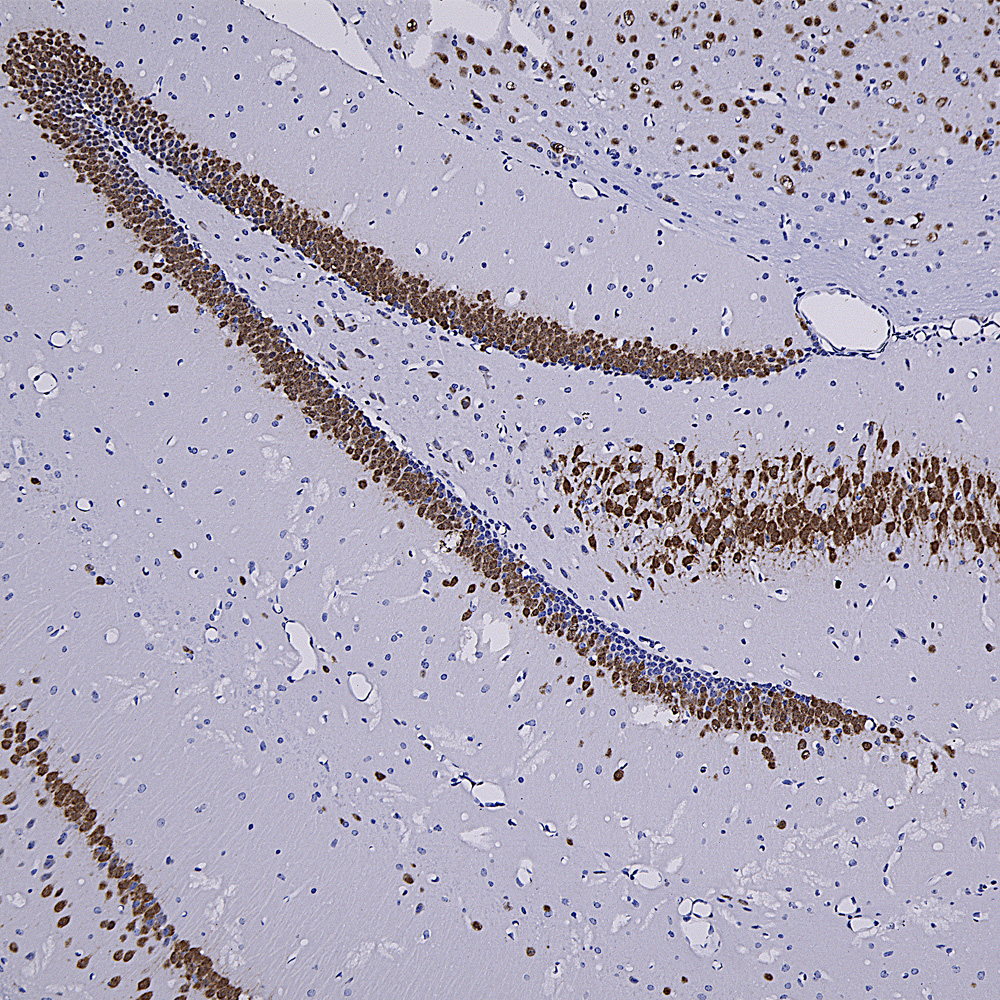
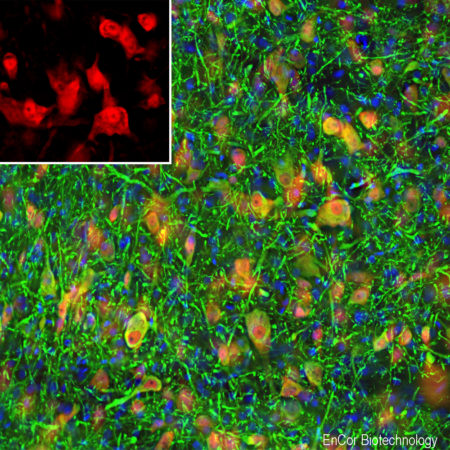
Chromogenic immunostaining of a formalin fixed paraffin embedded mouse hippocampus section with rabbit pAb to Fox3/NeuN, RPCA-FOX3, dilution 1:4,000, detected with DAB (brown) using the Vector Labs ImmPRESS method and reagents with citra buffer retrieval. The RPCA-FOX3 antibody selectively labels the nuclei and distal perikaryal of most neuronal cell populations. This antibody performs well in testing with 4% PFA and NBF fixed mouse, human, and rat tissues. Mouse select image for larger view.
1. Mullen RJ, Buck CR, Smith AM. NeuN, a neuronal specific nuclear protein in vertebrates. Development 116:201-11 (1994).
2. Hodgkin J, Zellan JD, Albertson DG. Identification of a candidate primary sex determination locus, fox-1, on the X chromosome of Caenorhabditis elegans. Development 120:3681-3689 (1994).
3. Kim KK, Adelstein RS, Kawamoto S. Identification of neuronal nuclei (NeuN) as Fox-3, a new member of the Fox-1 gene family of splicing factors. J. Biol. Chem. 284:31052-61 (2009).
4. Underwood JG, et al. Homologues of the Caenorhabditis elegans Fox-1 protein are neuronal splicing regulators in mammals. Mol. Cell Biol. 25:10005-16 (2005).
5. Dredge BK, Jensen KB. NeuN/Rbfox3 nuclear and cytoplasmic isoforms differentially regulate alternative splicing and nonsense-mediated decay of Rbfox2. PLoS One doi: 10.1371/journal.pone.0021585 (2011).
6. Herculano-Houzel S, Lent R. Isotropic fractionator: a simple, rapid method for the quantification of total cell and neuron numbers in the brain. J. Neurosci. 25:2518-2521 (2005).
7. Azevedo FA. et al. Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled-up primate brain. J. Comp. Neurol. 513:532-41 (2009).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com