| Name: | Rabbit polyclonal antibody to GAPDH |
| Immunogen: | Full length recombinant human GAPDH expressed in and purified from E. coli |
| HGNC Name: | GAPDH |
| UniProt: | P04406 |
| Molecular Weight: | 36kDa |
| Host: | Rabbit |
| Isotype: | |
| Species Cross-Reactivity: | Human, rat, mouse, cow, pig, horse, chicken |
| RRID: | AB_2572289 |
| Format: | Antibody is supplied as an aliquot of serum plus 5mM NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:30,000 IF/ICC and IHC: 1:2,000 |
| Storage: | Store at 4°C for short term, for longer term storage store at -20°C. Avoid freeze / thaw cycles. |
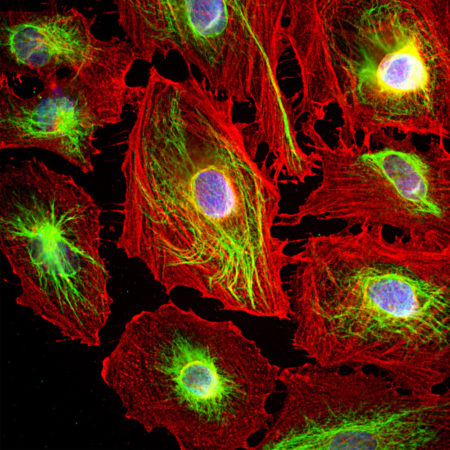
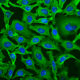
Immunofluorescent analysis of HeLa cells stained with rabbit pAb to GAPDH, RPCA-GAPDH, dilution 1:2,000 in red. Blue is Hoechst staining of nuclear DNA. The RPCA-GAPDH antibody produces diffuse cytoplasmic staining of cells.
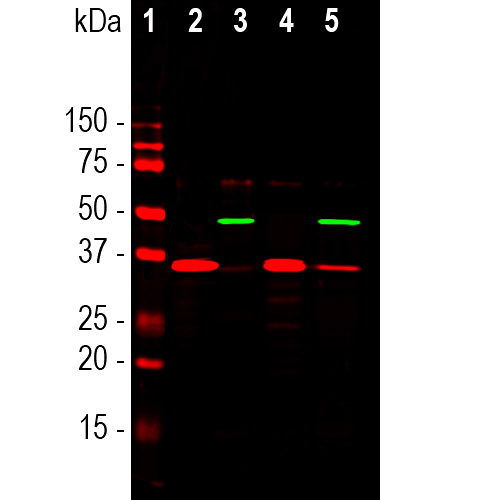
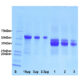
Western blot analysis of different cell cytosolic or nuclear enriched fractions, using rabbit pAb to GAPDH, RPCA-GAPDH, dilution 1:20,000, in red: [1] protein standard, [2] NIH-3T3 cytosolic, [3] NIH-3T3 nuclear, [4] HeLa cytosolic, and [5] HeLa nuclear fractions. Strong band at 37kDa corresponds to GAPDH protein, mainly detected in the cytosolic fractions. The same blot was simultaneously probed with mouse mAb to the nuclear RNA binding protein SF3B4, MCA-3A1, dilution 1:1,000, in green. In contrast to GAPDH, SF3B4 is exclusively expressed in the nuclear fraction.
Rabbit Polyclonal Antibody to GAPDH
Cat# RPCA-GAPDH
$120.00 – $800.00
Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is a metabolic enzyme responsible for catalyzing one step in the glycolytic pathway, the reversible oxidative phosphorylation of glyceraldehyde 3-phosphate to produce glyceraldehyde 1-3-bisphosphate. Because GAPDH protein is expressed in large amounts and is required at all times for an important “house keeping” function, levels of GAPDH mRNA are often measured and used as standards in studies of the expression of other mRNAs. Similarly specific antibodies to GAPDH are used to measure GAPDH expression as a protein standard in western blotting experiments. This antibody can be used as a loading control for such experiments, allowing comparison between the expression level of this protein and others in a cell or tissue. Apart from a role in glycolysis, GAPDH may have other roles, such as in the activation of transcription (1). GAPDH is also reported to bind to a variety of other proteins, including the amyloid precursor protein, mutations in which cause some forms of Alzheimer’s disease, and the polyglutamine tracts of Huntingtin, the protein product aberrant forms of which are causative of Huntington’s disease (2,3). The protein may also have a role in the regulation of apoptosis, and interestingly migrates from the cytoplasm into the nucleus when cells become apoptotic (4). The control of this is mediated by NO mediated S-nitrosylation of GAPDH in the cytoplasm which then binds to and stabilizes the Siah1 E3 ubiquitin ligase which enters the nucleus along with nitrosylated GAPDH. Nuclear Siah1 then initiates specific protein degradation events leading to the activation of a nuclear cell death pathway (5). Recently GAPDH was shown to be a substrate for the 5′-AMP dependent protein kinase (AMPK) which results in the redistribution of cellular GAPDH to cytosolic membranes and the inhibition of intracellular transport (6). AMPK is activated as AMP levels increase in response to starvation, and the pathway including GAPDH and transport inhibition apparently conserves cellular energy.
The immunogen used to raise RPCA-GAPDH was extensively purified pig GAPDH. The antibody has been widely used as a western blotting standard by many labs for many years and is known to detect GAPDH in a variety of mammalian species including human, rat and mouse. It produces a single clean band on western blots of typical cell and tissue extracts We also market a widely used mouse monoclonal antibody to GAPDH, MCA-1D4 with similar properties to RPCA-GAPDH. Mouse select image at left for larger view.
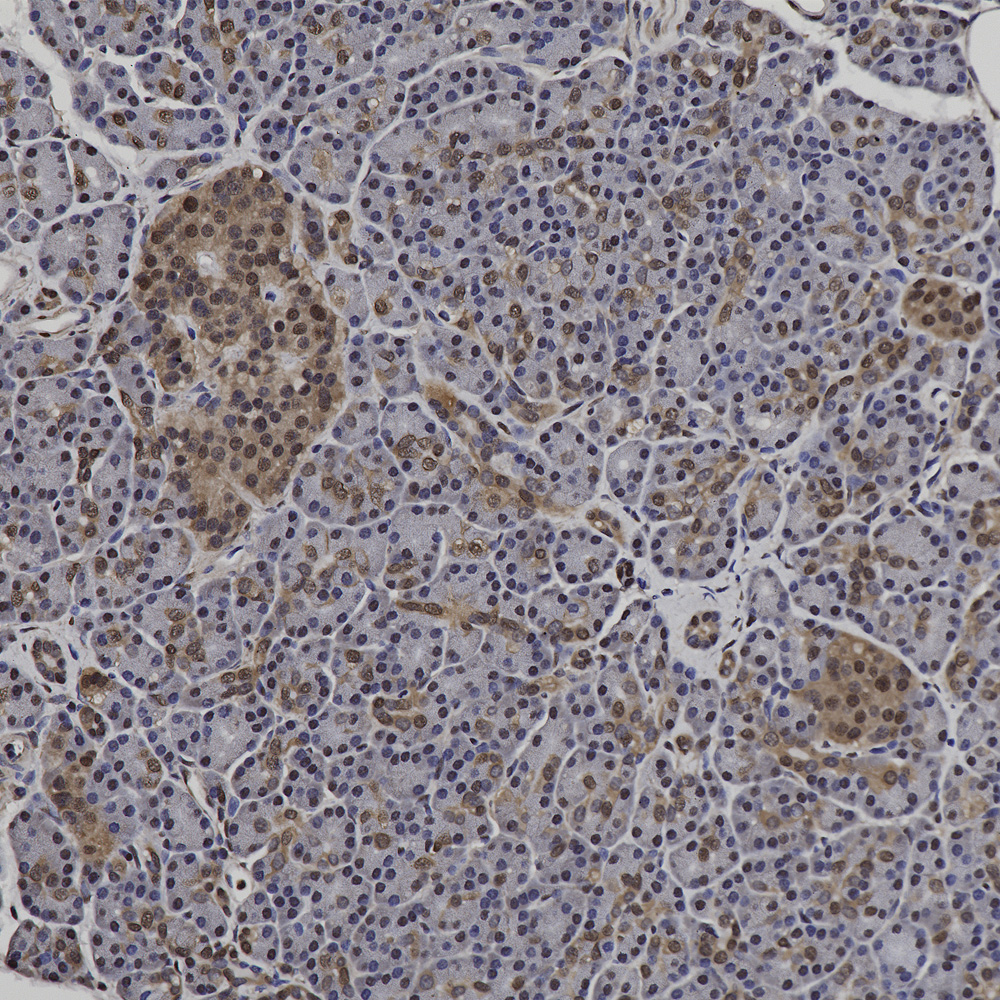
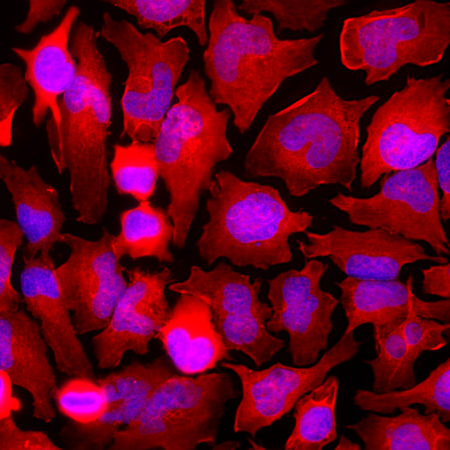
Chromogenic immunostaining of a NBF fixed paraffin embedded human pancreas section with rabbit pAb to GAPDH, RPCA-GAPDH, dilution 1:5,000, detected with DAB (brown) using the Vector Labs ImmPRESS method and reagents with citra buffer retrieval. Hematoxylin (blue) was used as the counterstain. The GAPDH antibody labels pancreatic exocrine glandular cells as well as a sub-population of endocrine cells. This antibody performs well in testing with both 4% PFA and standard NBF fixed human, mouse, and rat tissues. Mouse select image for larger view.
1. Morgenegg G, et al. Glyceraldehyde-3-phosphate dehydrogenase is a nonhistone protein and a possible activator of transcription in neurons. J. Neurochem. 47:54-62 (1986).
2. Schulze H, et al. Rat brain glyceraldehyde-3-phosphate dehydrogenase interacts with the recombinant cytoplasmic domain of Alzheimer’s beta-amyloid precursor protein. J Neurochem. 60:1915-22 (1993).
3. Burke JR, et al. Huntingtin and DRPLA proteins selectively interact with the enzyme GAPDH. Nature Med. 2:347-50 (1996).
4. Dastoor Z, Dreyer J-L. Potential role of nuclear translocation of glyceraldehyde-3-phosphate dehydrogenase in apoptosis and oxidative stress. J. Cell Sci. 114:1643-53 (2001).
5. Hara MR, et al. S-nitrosylated GAPDH initiates apoptotic cell death by nuclear translocation following Siah1 binding. Nat. Cell Biol. 7:665-74 (2005).
6. Yang J-S, et al. GAPDH inhibits intracellular pathways during starvation for cellular energy homeostasis. Nature 561:263-67 (2018).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com