| Name: | Mouse monoclonal to Parvalbumin |
| Immunogen: | Full-length recombinant human protein expressed in and purified from E. coli. |
| HGNC Name: | PVALB |
| UniProt: | P20472 |
| Molecular Weight: | 12kDa |
| Host: | Mouse |
| Isotype: | IgG1 |
| Species Cross-Reactivity: | Human, Rat, Mouse, Cow, Pig, Horse |
| RRID: | AB_2572372 |
| Format: | Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:1,000-1:5,000 ICC/IF or IHC: 1:1,000-1:5,000 |
| Storage: | Stable at 4°C for one year, for longer term store at -20°C |
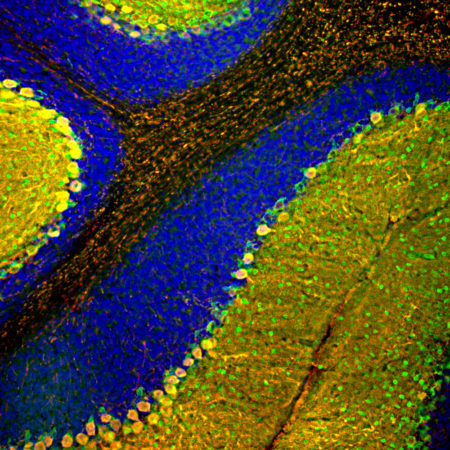
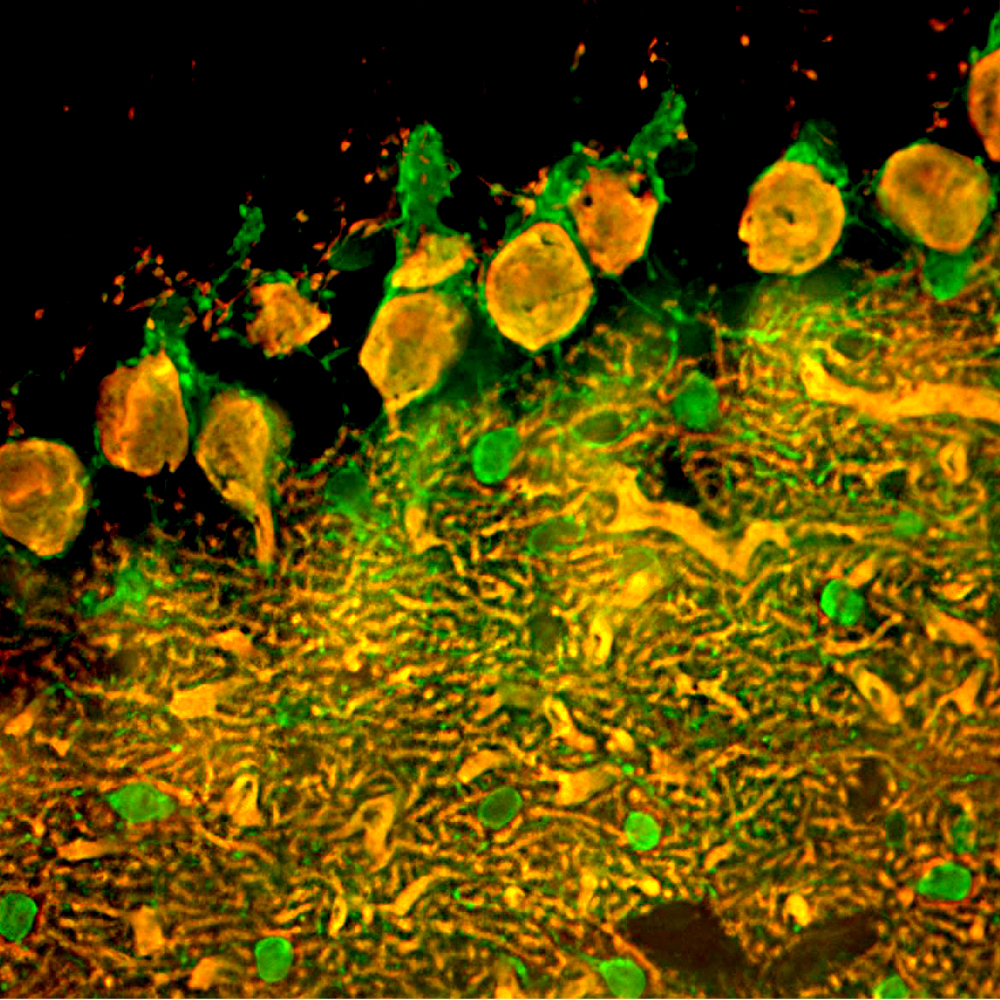
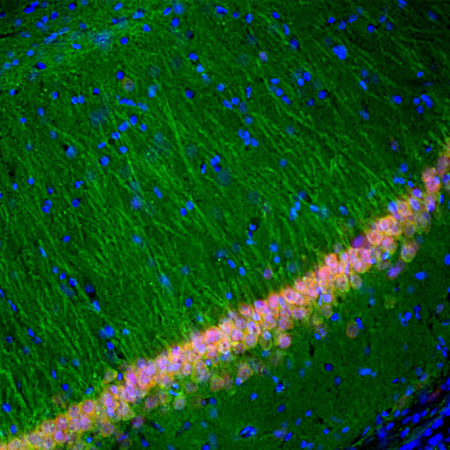
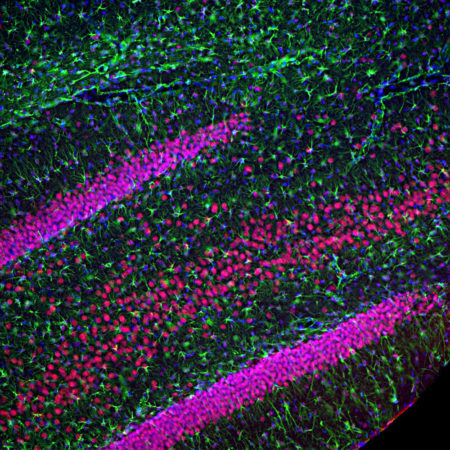
Immunofluorescent analysis of rat cerebellum section stained with mouse mAb to parvalbumin, MCA-3C9, dilution 1:1,000, in green, and costained with chicken pAb to calbindin, CPCA-Calb, dilution 1:2,000 in red. The blue is DAPI staining of nuclear DNA. Following transcardial perfusion of rat with 4% paraformaldehyde, brain was post fixed for 24 hours, cut to 45μM, and free-floating sections were stained with above antibodies. Most Purkinje cells strongly express both parvalbumin and calbindin and so appear yellow, whereas basket, stellate and Golgi cells express parvalbumin alone and so appear green.
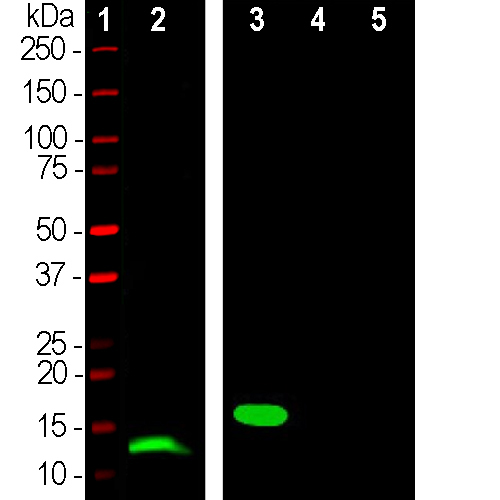
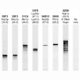
Western blot analysis of skeletal muscle lysates and His-tagged recombinant proteins using mouse mAb to parvalbumin, MCA-3C9, dilution 1:1,000 in green: [1] protein standard (red), [2] mouse muscle, [3] full length human parvalbumin, [4] full length human calretinin, and [5] full length human calbindin. A band at 12kDa is detected in in muscle lysate and one at 18kDa in the His-tagged recombinant parvalbumin protein lane as expected since the His-tag and other vector derived sequence adds about 6kDa to the molecule. Note that the MCA-3C9 antibody is not cross-reactive with either calbindin or calretinin despite their related amino acid sequences.
Mouse Monoclonal Antibody to Parvalbumin
Cat# MCA-3C9
$120.00 – $800.00
Parvalbumin is a low molecular weight cytoplasmic Calcium binding protein containing the “EF hand” Calcium binding motif and is the first protein characterized in this subclass (1). Parvalbumin is expressed in fast-contracting muscles, in the brain and in some endocrine tissues (2,3). In brain it is particularly concentrated in Purkinje cells and interneurons in the molecular layer, but is also found in many cortical GABAergic interneurons. These GABAergic interneurons in most cases express only one of three Calcium binding proteins, namely parvalbumin, calretinin or calbindin (4-6). Each type of interneuron has distinct electrophysiological properties and as a result, different types of interneuron can be identified and classified based on their content of these three proteins (7).
The MCA-3C9 antibody was made against full length recombinant human parvalbumin expressed in and purified from E. coli. Since parvalbumin is related in amino acid sequence to both calretinin and calbindin, we also expressed these proteins to check that our various reagents show no cross reactivity. So our antibodies to parvalbumin are useful cell type markers provided, as is the case with this antibody, they do no cross react with the related molecules calretinin or calbindin. The antibody works well for western blotting and for IF, ICC and IHC (for IHC see data under “Additional Info” tab). EnCor also manufactures a chicken polyclonal antibody to parvalbumin CPCA-Pvalb. We also supply a variety of other mouse and chicken antibodies to calbindin (MCA-5A9 and CPCA-Calb) and calretinin (MCA-3G9, MCA-6A9 and CPCA-Calret), allowing double and triple labeling of appropriate cell and tissue samples. Mouse select image at left for larger view.
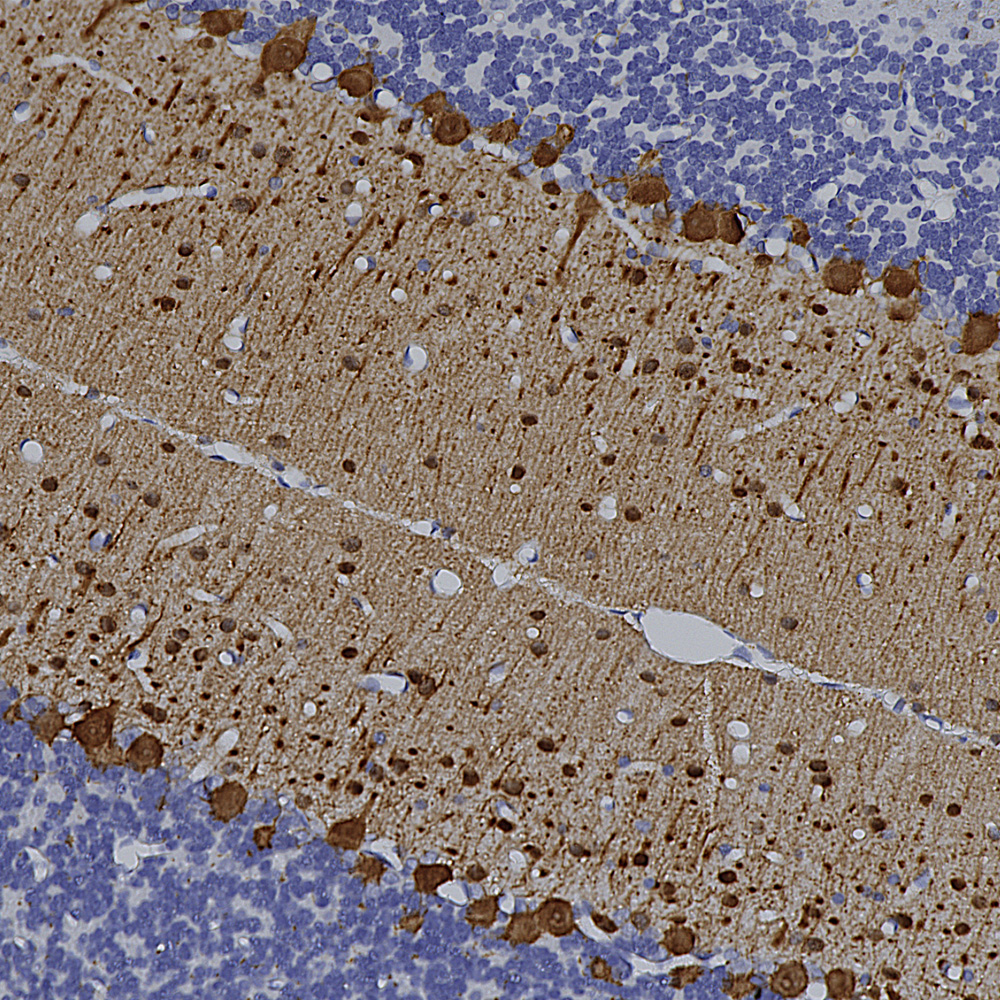
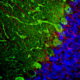
Chromogenic immunostaining of a 4% PFA fixed paraffin embedded rat cerebellum section with mouse mAb to parvalbumin, MCA-3C9, dilution 1:2,000, detected in DAB (brown) using the Vector Labs ImmPRESS method and reagents with citra buffer retrieval. Hematoxylin (blue) was used as the counterstain. In the cerebellum, MCA-3C9 antibody strongly labels Purkinje cells, basket cells and Golgi cells. This antibody performs well in testing with 4% PFA or standard NBF fixed rat and human tissues. Mouse select image for larger view.
Immunofluorescent analysis of rat brain cerebellum section stained with mouse mAb to parvalbumin, MCA-3C9, dilution 1:1,000, in green, and costained with chicken pAb to calbindin, CPCA-Calb, dilution 1:2,000 in red. Following transcardial perfusion of rat with 4% paraformaldehyde, brain was post fixed for 24 hours, cut to 45μM, and free-floating sections were stained with above antibodies. Most Purkinje cells coexpress parvalbumin and calbindin and so appear yellow, whereas basket, stellate and Golgi cells express parvalbumin only and so appear green. Mouse select image for larger view.
1. Kretsinger RH, Nockolds CE. Carp Muscle Calcium-binding Protein: II. Structure determination and general description. J. Biol. Chem. 248:3313-26 (1973).
2. Andressen C, Bliimcke I, Celio MR. Calcium-binding proteins: selective markers of nerve cells. Cell Tissue Res. 271:181-208 (1993).
3. Schwaller B, Meyer M, Schiffmann S. ‘New’ functions for ‘old’ proteins: The role of the calcium binding proteins calbindin D-28k, calretinin and parvalbumin, in cerebellar physiology. Studies with knockout mice. The Cerebellum 1:241–58 (2002).
4. Celio MR. Calbindin D-28k and parvalbumin in the rat nervous system. Neurosci. 35:375-475 (1990).
5. Condé F, et al. Local circuit neurons immunoreactive for calretinin, calbindin D‐28k or parvalbumin in monkey prefronatal cortex: Distribution and morphology. J. Comp. Neurol. 341:95-116 (1994).
6. Hof PR, et al. Cellular distribution of the calcium-binding proteins parvalbumin, calbindin, and calretinin in the neocortex of mammals: phylogenetic and developmental patterns. J. Chem. Neuroanat. 16:77-116 (1999).
7. Bearzatto B, et al. Mono- and dual-frequency fast cerebellar oscillation in mice lacking parvalbumin and/or calbindin D-28k. Eur. J. Neurosci. 22:861-70 (2005).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com