| Name: | Mouse monoclonal antibody to amyloid a-beta |
| Immunogen: | 1-42 human amyloid Aβ epitope is sequence 1-16 |
| HGNC Name: | APP |
| UniProt: | P05067 |
| Molecular Weight: | ~5kDa |
| Host: | Mouse |
| Isotype: | IgG2a heavy, κ light |
| Species Cross-Reactivity: | Human |
| RRID: | AB_2572226 |
| Format: | Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:1,000-1:2,000. IF/ICC: 1:1,000. IHC: 1:2,000. |
| Storage: | Stable at 4°C for one year, for longer term store at -20°C |
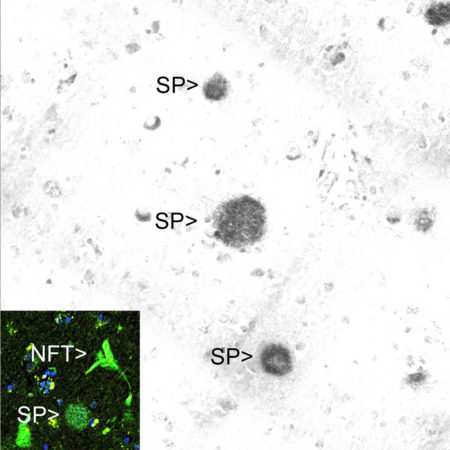
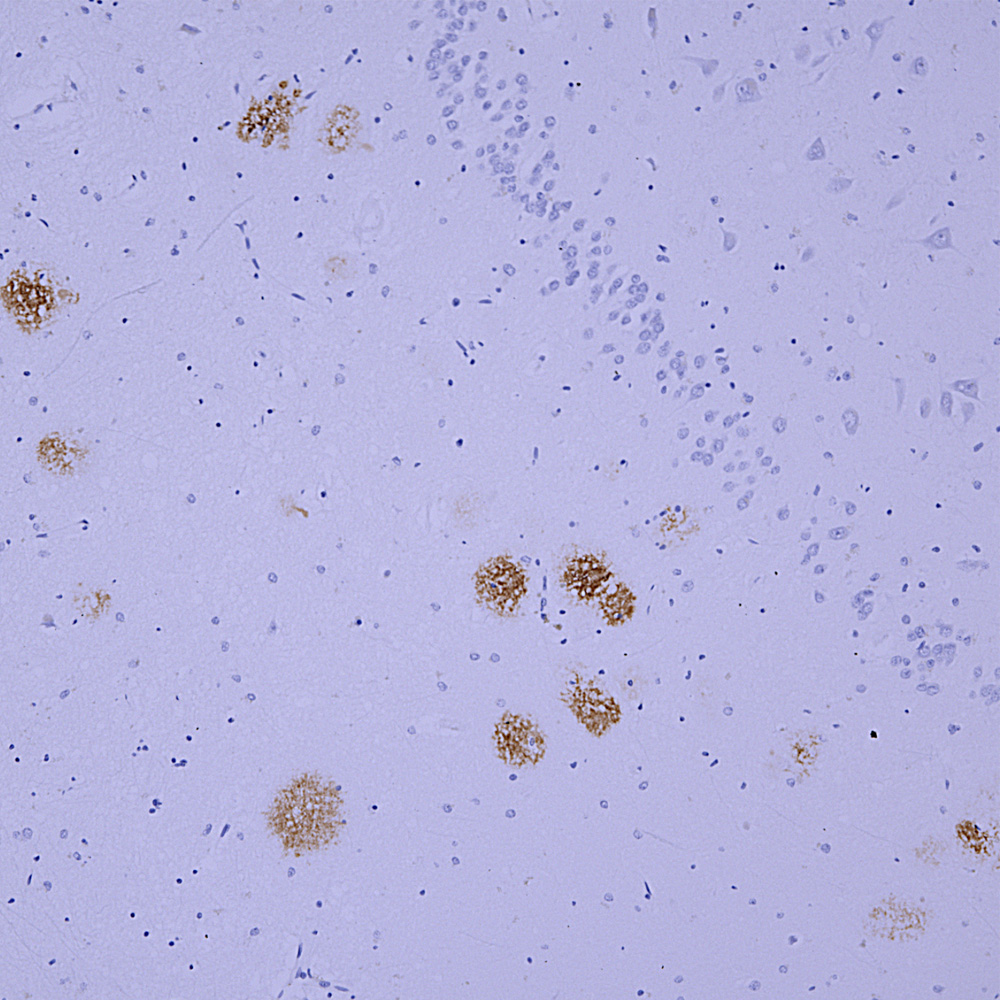
Immunohistochemical analysis of a region of cerebral cortex from an Alzheimer’s disease (AD) patient stained with MCA-AB9, the signal detected with a secondary anti-mouse antibody coupled to HRP, signal revealed with DAB. Senile plaques are labeled “SP”. The region of the lowest of the three plaques is shown in the inset stained with the fluorescent dye thioflavin-S. This dye binds to not only the senile plaque but also a neurofibrillary tangle (NFT), the other pathological hallmark of AD, which do not contain Aβ.
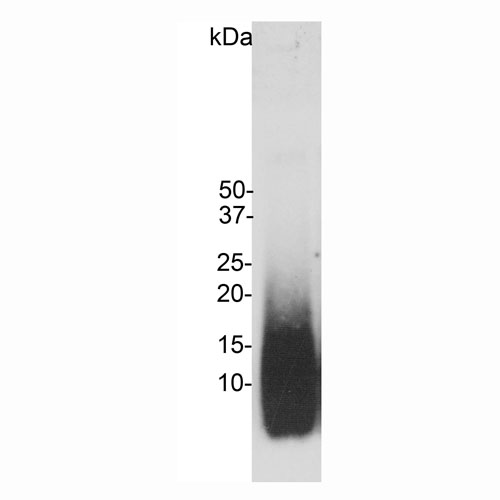
Blot of amyloid-β peptide preparation probed with MCA-AB9. The MCA-AB9 antibody recognizes monomeric amyloid-β peptide running at ~5kDa and also higher molecular weight amyloid-β aggregates.
Mouse Monoclonal Antibody to Amyloid Aβ
Cat# MCA-AB9
$120.00 – $800.00
Alzheimer’s disease (AD) is a serious and increasingly common age related dementia which is characterized by the formation of senile plaques which are extracellular accumulations of insoluble proteins. Another characteristic of AD is the formation of neurofibrillary tangles inside neurons. A major component of the senile plaques is β-amyloid, a.k.a. Aβ, a peptide the predominant forms of which are 42 or 40 amino acids in length. The Aβ peptide is derived by proteolytic cleavage from a much larger protein membrane lozalized called the amyloid precursor protein (APP). Deposition of Aβ is increased as a result of certain point mutations in the APP gene and by mutations in presenilins and other genes. The presence of certain alleles of the ApoE gene also increase the likelihood of progressing to AD.
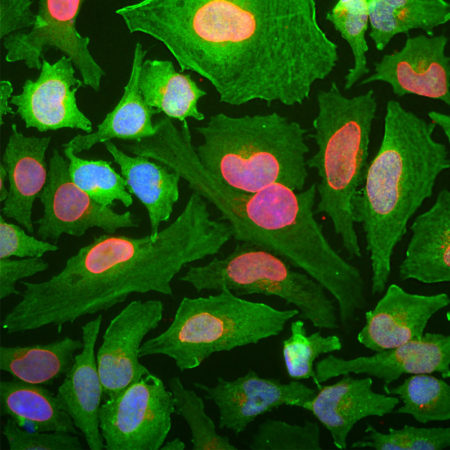
The MCA-AB9 is a mouse monoclonal antibody which made against the Aβ peptide and has been described in a peer reviewed publication (1). It was found to bind to the N-terminal peptide of Aβ, and has become very widely used (e.g. 2-6). It works well on western blots and reveals senile plaques in AD brains by ICC and IHC, for further IHC see data under “Additional Info” tab. Much interest has focused on the use of humanized monoclonal antibodies to target and potentially clear Aβ from the brains of Alzheimer patients. This antibody has been used in this manner as a proof of concept in mouse models of AD (e.g. 2,3). This antibody was originally made in the Mayo Clinic in Jacksonville, Florida, in the then laboratory of Dr. Todd Golde. Mouse select image at left for larger view.
Chromogenic immunostaining of a NBF fixed paraffin embedded human hippocampus section from an Alzheimer’s Disease case. Mouse mAb to amyloid αβ, MCA-AB9 dilution 1:2,000, was detected in DAB (brown) following the ImmPress method with citra buffer retrieval. Hematoxylin (blue) was used as the counterstain. MCA-AB9 specifically labels the amyloid plaques characteristic of this disease as identified by their morphology. This antibody performs well in standard NBF fixed tissues but has not been tested in other fixatives or species. Mouse select image for larger view.
1. Levites Y, et al. Anti-Abeta42- and anti-Abeta40-specific mAbs attenuate amyloid deposition in an Alzheimer disease mouse model, J. Clin. Invest 116:193-201 (2006).
2. Wang A, et al. Robust Amyloid Clearance in a Mouse Model of Alzheimer’s Disease Provides Novel Insights into the Mechanism of Amyloid-β Immunotherapy. J. Neuroscience, March 16, 2011 • 31:4124–36 (2011).
3. Kim J, et al. Aβ40 Inhibits Amyloid Deposition In Vivo. J. Neurosci. 27:627-33 (2007).
4. Das P, et al. Amyloid-β Immunization Effectively Reduces Amyloid Deposition in FcRγ-/- Knock-Out Mice. J Neurosci. 23:8532-8 (2003).
5. Sagi SA, et al. Substrate Sequence Influences γ-Secretase Modulator Activity: Role of the Transmembrane Domain of The Amyloid Precursor Protein. J. Biol. Chem. 286:39794-803 (2011).
6. Fernandez-Funez P, et al. Holdase activity of secreted Hsp70 masks amyloid-β42 neurotoxicity in Drosophila. 2016.
7. Moore BD, et al. Overlapping profiles of Aβ peptides in the Alzheimer’s disease and pathological aging brains. Alz. Res. Therap. 4:18 (2012).
This antibody has been utilized in numerous other peer-reviewed publications many of which can be found by searching Google Scholar for “AB9 AND Amyloid” or by selecting here.
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com