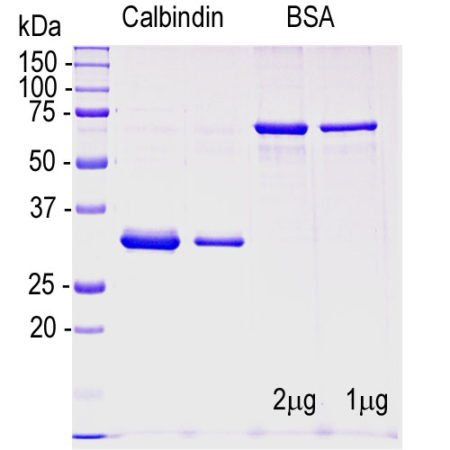
A codon optimized cDNA encoding full length human calbindin 1 was generated and inserted into the pET29a (+) expression vector. The vector adds an C-terminal His-tag to the human sequence which increases the molecular weight by about 5kDa. The construct was expressed by standard methods in E. coli and purified using a Nickel column in 6M urea, and the protein is supplied in this form. The lane on the far left contains protein standards of the indicated molecular size. In the next lanes 2µg and 1µg of the recombinant human calbindin 1 were run as indicated and in the next two lanes show 2µg and 1µg of BSA.
Human Calbindin, Cat# Prot-r-Calb1
$300.00 – $2,000.00
Calbindin-1, also known as calbindin-D28k or just as calbindin, is a member of the superfamily of cytoplasmic Ca2+-binding proteins. Calbindin-1 is expressed in the mammalian brain, intestine, kidney and pancreas. In the brain it is localized in certain classes of neurons, and antibodies to it are useful for identifying specific neuronal cell types. It is particularly concentrated in the dendrites and perikarya of cerebellar Purkinje cells, but is also found in many GABAergic interneurons in the cortex. These GABAergic interneurons in most cases express only one of three Ca2+ binding proteins, namely calbindin or parvalbumin or calretinin. As a result these important inhibitory interneurons can be subclassified and identified based on their content of these proteins.
Calbindin-1, also known as calbindin-D28k or just as calbindin, is a member of the superfamily of cytoplasmic Ca2+-binding proteins. Calbindin-1 belongs to the subclass of these containing the “EF hand” Ca2+ binding motif originally characterized in parvalbumin (1). This type of Ca2+ binding motif is distinct from the “pseudo EF hand” motif characteristic of the very large S100family of Ca2+ binding proteins. Calbindin-1 is expressed in the mammalian brain, intestine, kidney and pancreas. In the brain it is localized in certain classes of neurons, and antibodies to it are useful for identifying specific neuronal cell types (2). It is particularly concentrated in the dendrites and perikarya of cerebellar Purkinje cells, but is also found in many GABAergic interneurons in the cortex. These GABAergic interneurons in most cases express only one of three Ca2+ binding proteins, namely calbindin or parvalbumin or calretinin. As a result these important inhibitory interneurons can be subclassified and identified based on their content of these proteins (2). Each type of neuron as defined in this fashion has particular electrophysiological and functional properties. For example, calbindin-1 positive interneurons are not fast-spiking as are parvalbumin expressing interneurons. Human calbindin-1 is a 263 amino acid protein with an SDS-PAGE weight close to the actual molecular weight of 28kDa. It is related in primary sequence to calretinin, which is also known as 29 kDa calbindin and calbindin-2. Our construct includes the entire human calbindin-1 sequence and has an additional ~5 kDa due to the His-tag and so runs at about 33 kDa on SDS-PAGE. The His-tag and other vector derived sequence can be removed if required. The primary sequence and NMR structure of calbindin-1 indicates six distinct Ca2+-binding sites corresponding to the EF hands motifs. Of the six sites four bind Ca2+ with relatively high affinity. The function of calbindin-1 appears to be primarily buffering the Ca2+ level in cells. The affinity of calbindin-1 for Ca2+ is low at the typical resting cytoplasmic Ca2+ level of around 100nM, and the protein only binds Ca2+ significantly when levels increase greatly. Accordingly it is widely thought that the primary function of this protein is to act as a Ca2+ buffer. Buffering Ca2+ is important, as uncontrolled increases in the level of this ion can lead to both apoptosis due to Ca2+ stimulated release of proteins from mitochondria and necrosis due to the activation of Ca2+ dependent proteases. Knockout of the calbindin-1 gene in mice leads to ataxia and other motor problems, consistent with the large amounts of protein normally present in the cerebellum (3).
1. Kretsinger RH, Nockolds CE. Carp Muscle Calcium-binding Protein: II. Structure determination and general description. J. Biol. Chem. 248:3313-3326 (1973).
2. Andressen C, Bliimcke I, Celio MR. Calcium-binding proteins: selective markers of nerve cells. Cell Tissue Res 271:181-208 (1993).
3. Schwaller B, Meyer M, Schiffmann S. ‘New’ functions for ‘old’ proteins: The role of the calcium binding proteins calbindin D-28k, calretinin and parvalbumin, in cerebellar physiology. Studies with knockout mice. The Cerebellum 1:241–258 (2002).
Related products
Contact info
EnCor Biotechnology Inc.
4949 SW 41st Boulevard, Ste 40
Gainesville
Florida 32608 USA
Phone: (352) 372 7022
Fax: (352) 372 7066
E-mail: admin@encorbio.com