

EnCor Biotechnology
Mouse Monoclonal Antibody to Aldolase C (ALDOC, ALDC, Zebrin II), Cat# MCA-1A1
Description
The MCA-1A1 antibody was made against recombinant human aldolase C C-terminal peptide KYEGSGEDGGAAAQSLYIANHAY, amino acids 342-364 of the NCBI sequence 1XFB_A. We then used nested 20 amino acid peptides to map the MCA-1A1 epitope to the peptide KYEGSGEDGGA, amino acids 342-352 of the human aldolase C sequence. Interestingly the antibody does not bind mouse aldolase C, which is somewhat different in sequence, RYEGSGDGGA, with a R>K change and missing an E residue (see here). Since MCA-1A1 binds to aldolases from other species also lacking the E residue and since the K>R change is unique to mouse we can conclude that the K342 is essential for MCA-1A1 binding. We also recommend our alternative mouse antibody MCA-4A9, which binds the N-terminal peptide of the human molecule and on western blots binds to all mammalian aldolase C tested. A third EnCor mouse monoclonal antibody MCA-E9, binds a region in the center of aldolase C which is virtually identical in aldolase A, B and C and consequently, as would be expected, it binds all three proteins. MCA-E9 is not recommended for IF and IHC applications.
- Cell Type Marker
- Cytoplasmic Enzyme
- Epitope Mapped Antibodies
- Immunohistochemistry Verified
- Mouse Monoclonal Antibodies
Add a short description for this tabbed section
| Immunogen: | C-terminal sequence KYEGSGEDGGAAAQSLYIANHAY 342-364 in NCBI sequence 1XFB_A |
| HGNC Name: | ALDOC |
| UniProt: | P09972 |
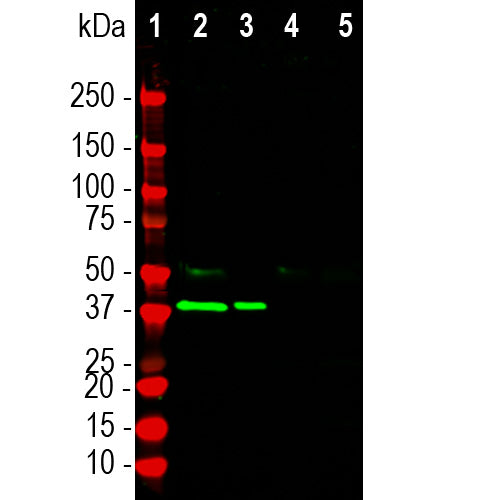
| Molecular Weight: | ~40kDa |
| Host: | Mouse |
| Isotype: | IgG1 |
| Species Cross-Reactivity: | Human, rat, dog, cow, not mouse |
| RRID: | AB_2572224 |
| Format: | Protein G affinity purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IF, ICC, IHC |
| Recommended Dilutions: | WB: 1:2,000. IF: 1:5,000. ICC: 1:1,000. IHC: 1:5,000-1:10,000. |
| Storage: | Store at 4°C for short term, for longer term store at -20°C. Stable for 12 months from date of receipt. |
Aldolases are important glycolytic cytosolic enzymes which catalyse the reversible conversion of fructose-1,6-bisphosphate to glyceraldehyde 3-phosphate. There are three aldolase isozymes coded by three distinct genes in mammals, namely aldolase A, B, and C. Aldolase A is heavily expressed in muscle enzyme and aldolase B is heavily expressed in liver and is regarded as a liver specific enzyme (1-5). In the adult, aldolase C is the brain-specific isozyme expressed in astrocytes and a few classes of neurons, notably Purkinje cells (4). Appropriate antibodies to aldolase C are therefore useful to identify astrocytes in cell culture and sectioned material, and the enzyme may be over expressed in some forms of cancer (6). Recent studies also suggest that detection of aldolase C in blood may be a useful biomarker of the severity and recovery process in patients suffering from the sequelae of traumatic brain injury (7). AldC corresponds to the protein Zebrin II, originally characterized by Brochu et al. (10). Zebrin II was characterized as an initially unknown antigen present in bands of Purkinje cells alternating with bands of Purkinjes which were Zebrin II negative.

Immunofluorescent staining of a rat cerebellum section with MCA-1A1, diluted 1:5,000 in red and rabbit antibody to GFAP, RPCA-GFAP, 1:5,000 in green. The MCA-1A1 antibody reveals the perikarya and dendrites of certain Purkinke cells in the molecular layer. Hoechst stain shows nuclei of all cell types in blue.

Chromogenic immunostaining of a 4%PFA fixed paraffin embedded rat cerebellum section with mouse mAb to Aldolase C, MCA-1A1, dilution 1:10,000, detected with DAB (brown) using the Vector Labs ImmPRESS method and reagents with citrate buffer retrieval. Hematoxylin (blue) was used as the counterstain. The MCA-1A1 antibody strongly labels the cell bodies and dendrites of certain Purkinje cells but fails to stain others. This antibody performs well in testing with both 4% PFA and standard NBF fixed tissues. Mouse select image for larger view.
1. Popovici T, et al. Localization of aldolase C mRNA in brain cells. FEBS Lett. 268:189-193 (1990).
2. Weber A, et al. Dietary Control of Aldolase B and L-type Pyruvate Kinamse RNAs in Rat. J. Biol. Chem. 259:1798-802 (1984).
3. Mukai T, et al. The structure of the brain-specific rat aldolase C gene and its regional expression. BBRC 174:1035-42 (1991).
4. Royds J, et al. Monoclonal antibody to aldolase C: a selective marker for Purkinje cells in the human cerebellum. Neuropathol. Appl. Neurobiol. 13:11-21 (1987).
5. Thompson R., Kynoch P. Willson V. Cellular localization of aldolase C subunits in human brain. Brain Res. 232:489-93 (1982).
6. Schapira F, Reuber M, Hatzfeld A. Resurgence of two fetal-type of aldolases (A and C) in some fast-growing hepatomas. BBRC 40:321-27 (1970).
7. Arai Y, et al. Position-independent, high-level, and correct regional expression of the rat aldolase C gene in the central nervous system of transgenic mice. Eur. J. Biochem. 221:253-260 (1994).
8. Wachsmuth E, Thorner M, Pfleiderer G. The cellular distribution of aldolase isozymes in rat kidney and brain determined in tissue sections by the immuno-histochemical method. Histochem. 45:143-61 (1975).
9. Walther EU, et al. Genomic sequences of aldolase C (Zebrin II) direct lacZ expression exclusively in non-neuronal cells of transgenic mice. PNAS 95:2615-20 (1998).
10. Brochu U et al. Zebrin II: a polypeptide antigen expressed selectively by Purkinje cells reveals compartments in rat and fish cerebellum J.Comp. Neurol. 291:538-552 (1990).
Add a short description for this tabbed section