EnCor Biotechnology
Mouse Monoclonal Antibody to αII-Spectrin Cat# MCA-3D7
Description
The MCA-3D7 antibody was made against a recombinant human protein construct derived from the C-terminus of αII-spectrin comprising the C-terminal 2 spectrin repeats, specifically amino acids 2086-2476 from AAB41498.1 expressed in and purified from E. coli. This antibody can be used to study αII-spectrin on western blots, including to visualize the 150kDa and 145kDa calpain breakdown products and the 150kDa and 120kDa caspase breakdown products. It can also be used to image the neuronal plasma membrane cytoskeleton in cells in culture. We also make a rabbit polyclonal antibody with similar properties, RPCA-aII-Spec.
- Cell Structure Marker
- Cell Type Marker
- Cytoskeletal Marker
- Epitope Mapped Antibodies
- Mouse Monoclonal Antibodies
Add a short description for this tabbed section
| Name: | Alpha-II-spectrin, mouse monoclonal antibody, Cat# MCA-3D7 |
| Immunogen: | Recombinant C-terminal region of human αII spectrin expressed in and purified from E. coli |
| HGNC Name: | SPTAN1 |
| UniProt: | Q13813 |
| Molecular Weight: | ~240kDa |
| Host: | Mouse |
| Isotype: | IgG1 |
| Species Cross-Reactivity: | Human, Rat, Mouse |
| RRID: | AB_2572381 |
| Format: | Purified antibody at 1mg/mL in 50% PBS, 50% glycerol plus 5mM NaN3 |
| Applications: | WB, IF/ICC, IHC |
| Recommended Dilutions: | WB: 1:3,000. IF/ICC: 1:500. |
| Storage: | Store at 4°C for short term, for longer term store at -20°C. |
The spectrin family of proteins was originally discovered as the major components of the submembranous cytoskeleton of osmotically lysed red blood cells (1). The lysed blood cells could be seen as transparent red blood cell shaped objects in the light microscope and were referred to as red cell “ghosts”. The major proteins of these “ghosts” proved to be actin, ankyrin, band 4.1, and several other proteins including two major bands appearing as 240kDa and 260kDa bands on SDS-PAGE gels. This pair of proteins was named “spectrin” since they were discovered in the red blood cell “ghosts” (1). Later work showed that similar high molecular weight bands were seen in membrane fractions from other eukaryotic cell types.
Work by Levine and Willard described a pair of ~240-260 kDa molecular weight proteins which weretransported at the slowest rate along mammalian axons (2). They named these proteins “fodrin” as antibody studies showed that they were localized in the sheath under the axonal membrane, but not in the core of the axon (fodros means sheath in Greek). Subsequently, fodrin was found to be a member of the spectrin family of proteins, and the spectrin nomenclature is now normally used (3).
Spectrins form tetramers of two α and two β subunits, with the α corresponding to the lower molecular weight ~240 kDa band and the β corresponding to the ~260 kDa or in some case much larger band. Most spectrin tetramers are about 0.2 microns or 200 nm long, and each α and β subunit has a cell type-specific expression pattern. The basic structure of each spectrin subunit is the spectrin repeat, which is a sequence of about 110 amino acids which defines a compact domain containing three closely packed α-helices. Each spectrin subunit contains multiple copies of this repeat, with 20 in each of the α subunits. The β I-IV subunits each contain 17 spectrin repeats, while the β V subunit, also known as β-heavy spectrin, contains 30 of these repeats. The various subunits also contain several other kinds of functional domains, allowing the spectrin tetramer to interact with a variety of protein, ionic, and lipid targets. The α-subunits each contain one calmodulin-like calcium binding region and one Src-homology 3 (SH3) domain, an abundant domain involved in specific protein-protein interactions. The β subunits all have a N-terminal actin-binding domain and may also have one SH3 domain and one pleckstrin homology domain, a multifunctional type of binding domain which in β-I spectrin at least binds the membrane lipid PIP2.
Spectrins are believed to have a function in giving mechanical strength to the plasma membrane since the tetramers associate with each other to form a dense submembranous geodesic meshwork (3). They also bind a variety of other membrane proteins and membrane lipids, and the proteins they bind to are therefore themselves localized in the membrane. Diseases may be associated with defects in one or other of the spectrin subunits (5). For example, some forms of hereditary spherocytosis, the presence of spherical red blood cells which are prone to lysis, can be traced to mutations in some of the spectrin subunits (6). The αII subunit is widely expressed in tissues but, in the nervous system, is found predominantly in neurons. Since the immunogen used to generate the antibody is at the C-terminus of the molecule, the antibody will reveal the 150kDa, 145kDa and 120kDa breakdown products generated by calpain and caspase cleavage on western blots (7). This antibody can also be used to identify neurons and fragments derived from neuronal membranes in cells, in tissue culture, and in sectioned material.

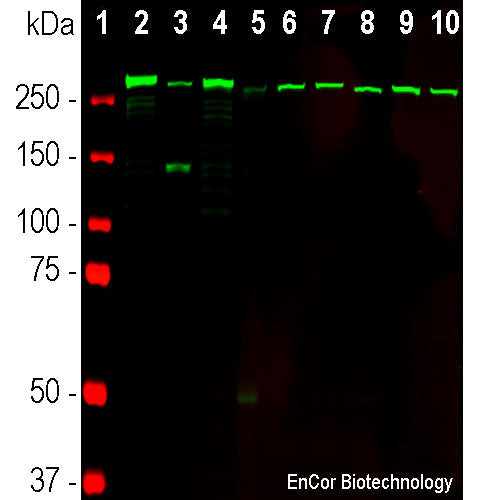
Western blot of SH-SY5Y cell homogenates probed with 1:500 MCA-3D7 from control cells in lane 1, from cells treated with 5nM maitotoxin for 2 hours in lane 2 and with 0.5mM staurosporin for 5 hours in lane 3. Maitotoxin increases cytoplasmic levels of Calcium, resulting in calpain activation, while staurosporin is a protein kinase inhibitor which induces apoptosis and hence caspase activation. All three lanes show intact α-II spectrin at ~286kDa. Lane 2 also shows the 150kDa caspase breakdown product while lane 3 also shows the 120kDa and in this case rather faint 145kDa calpain breakdown products.

Chromogenic immunostaining of a 4% PFA fixed paraffin embedded rat cerebellum section with mouse mAb to αII-spectrin, MCA-3D7, dilution 1:200, detected in DAB (brown) following the detected with DAB (brown) using the Vector Labs ImmPRESS method and reagents with citra buffer retrieval. Hematoxylin (blue) was used as the counterstain. The αII-spectrin antibody labels the cytoplasm and membranes of neurons, localizing to vesicles and plasma membranes. In the cerebellum, MCA-3D7 reveals the dendrites in the molecular layer and note the plasma membrane staining of granule neurons in the granule cell layer. This antibody performs well in testing with 4% PFA perfusion fixed tissue but does not work in overly fixed rodent or human tissues. Mouse select image for larger view.
1. Marchesi VT, Steers E. Selective solubilization of a protein component of the red cell membrane. Science 159:203-4 (1968).
2. Levine J, Willard M. Fodrin: axonally transported polypeptides associated with the internal periphery of many cells. J. Cell Biol. 90:631-42 (1981).
3. Bennett V, Baines AJ. Spectrin and ankyrin-based pathways: metazoan inventions for integrating cells into tissues. Physiol. Rev. 81:1353-92 (2001).
4. Djinovic-Carugo K, Gautel M, Ylänne J, Young P. The spectrin repeat: a structural platform for cytoskeletal protein assemblies. FEBS Lett. 513:119-23 (2002).
5. Bennett V, Healy J. Organizing the fluid membrane bilayer: diseases linked to spectrin and ankyrin. Trends Mol. Med. 14:28-36 (2008).
6. Eber S, Lux SE. Hereditary spherocytosis–defects in proteins that connect the membrane skeleton to the lipid bilayer. Semin. Hematol. 41:118-41 (2004).
7. Mondello S, et al. αII-spectrin breakdown products (SBDPs): diagnosis and outcome in severe traumatic brain injury patients J. Neurotrauma 27:1203-13 (2010).
Add a short description for this tabbed section